The following information was reported in the December 31 financial statements of National Airways, Incorporated (listed...
Fantastic news! We've Found the answer you've been seeking!
Question:
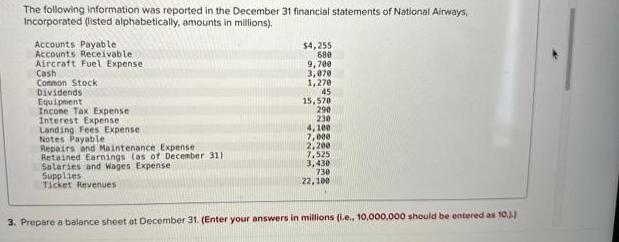
Transcribed Image Text:
The following information was reported in the December 31 financial statements of National Airways, Incorporated (listed alphabetically, amounts in millions). Accounts Payable. Accounts Receivable. Aircraft Fuel Expense Cash Common Stock Dividends Equipment Incone Tax Expense Interest Expense Landing Fees Expense Notes Payable Repairs and Maintenance Expense Retained Earnings (as of December 31) Salaries and Wages Expense Supplies Ticket Revenues $4,255 680 9,700 3,070 1,270 45 15,570 290 230 4,100 7,000 2,200 7,525 3,430 730 22,100 3. Prepare a balance sheet at December 31. (Enter your answers in millions (i.e., 10,000,000 should be entered as 10.) The following information was reported in the December 31 financial statements of National Airways, Incorporated (listed alphabetically, amounts in millions). Accounts Payable. Accounts Receivable. Aircraft Fuel Expense Cash Common Stock Dividends Equipment Incone Tax Expense Interest Expense Landing Fees Expense Notes Payable Repairs and Maintenance Expense Retained Earnings (as of December 31) Salaries and Wages Expense Supplies Ticket Revenues $4,255 680 9,700 3,070 1,270 45 15,570 290 230 4,100 7,000 2,200 7,525 3,430 730 22,100 3. Prepare a balance sheet at December 31. (Enter your answers in millions (i.e., 10,000,000 should be entered as 10.)
Expert Answer:
Answer rating: 100% (QA)
Certainly To prepare the balance sheet well organize the provided inf... View the full answer

Related Book For 

Fundamentals of Financial Accounting
ISBN: 978-0078025372
4th edition
Authors: Fred Phillips, Robert Libby, Patricia Libby
Posted Date:
Students also viewed these accounting questions
-
The following information was reported in the December 31, 2012, financial statements of Southwest Airlines, Inc. (listed alphabetically, amounts in millions). 1. Prepare an income statement for the...
-
The following information was reported in the December 31, 2017, financial statements of National Airways, Inc. (listed alphabetically, amounts in millions). 1. Prepare an income statement for the...
-
The following information was reported in the December 31, 2010, financial statements of Southwest Airlines, Inc. (listed alphabetically, amounts in millions). 1. Prepare an income statement for the...
-
Solve the linear equation with the x-intercept method. Check your answer. Approximate the solution to the nearest thousandth whenever appropriate. -1-x = = 0
-
What are convertible bonds? Why would a company issue convertible debt?
-
What are the four basic assumptions of robust optimization with independent parameters?
-
The thermal efficiency of gas turbine as compared to diesel engine is (a) higher (b) lower (c) same (d) unpredictable.
-
Ethanol is produced commercially by the hydration of ethylene: C2H4 (g) + H2O (v) C2H5OH (V) some of the product is converted to diethyl ether in the undesired side reaction 2 C2H5OH (v) = (C2H5)2O...
-
HISTORY OF PSYCHOLOGY PART 1 : Phrenology was a wildly popular but untrue scientific theory. Find another popular but untrue scientific theory. Find a URL on the internet exploring at least one more...
-
Tuckered Outfitters plans to market a custom brand of packaged trail mix. The ingredients for the trail mix will include Raisins, Grain, Chocolate Chips, Peanuts, and Almonds costing, respectively,...
-
There are five seats on the board of directors of Serbone Equity that are up for election. The firm has 320,400 shares of stock outstanding and uses straight voting. Each share is granted one vote...
-
The output voltage of a voltage divider is not fixed; it varies according to the load. a. Find the output voltage \(v_{\mathrm{o}}\) in Figure 6.13 for two different values of load resistance: (1)...
-
A pyridine-water solution, \(50 \mathrm{wt} \%\) pyridine, is to be continuously and countercurrently extracted at the rate of \(2.25 \mathrm{~kg} / \mathrm{s}\) with pure chlorobenzene to reduce the...
-
If \(1000 \mathrm{~kg} / \mathrm{h}\) of a cottonseed oil-oleic acid solution containing \(25 \mathrm{wt} \%\) acid is to be continuously separated into two products containing 2 and \(90 \mathrm{wt}...
-
The mathematical model of a nonlinear system is provided. Using the procedure outlined in this section, find the operating point and derive the linearized model. \(\begin{cases}\dot{x}_{1}=-x_{1}+3...
-
The mathematical model of a nonlinear system is provided. Using the procedure outlined in this section, find the operating point and derive the linearized model....
-
Mr. Thomas commenced business on 1 March 2022. During the first month of the operation, the following events and transactions occurred. Date 1 4 14 25 28 28 29 Business Transactions Started business...
-
The Strahler Stream Order System ranks streams based on the number of tributaries that have merged. It is a top-down system where rivers of the first order are the headwaters (aka outermost...
-
Assume you work as an assistant accountant in the head office of a DVD movie kiosk business, similar to Coinstar, Inc. With the increasing popularity of online movie rental operations, your company...
-
Match the type of inventory with the type of business by placing checkmarks in the applicable columns: Type of Business Type of Inventory Merchandising Manufacturing Merchandise Finished goods Work...
-
Rocktown Corporation bought 600 shares of General Eccentric stock on March 20, 2013, for its trading securities portfolio at $29 per share. Rocktown sold the stock at $33 per share on June 23, 2013....
-
How much heat is to be added to convert \(4 \mathrm{~kg}\) of water at \(20^{\circ} \mathrm{C}\) in to steam at 8 bar and \(200^{\circ} \mathrm{C}\). Take \(C_{p}\) of superheated steam as \(2.1...
-
What do you understand by mechanical and thermal efficiency? A steam plant uses 3 tonne of \(\mathrm{coal} / \mathrm{h}\). The steam is fed to turbine the output of which is \(4 \mathrm{MW}\). The...
-
Determine the total heat content per unit mass at the following state using the steam tables. Assume ambient pressure to be \(100 \mathrm{kPa}\) and \(C_{P}\) \(=2.0934 \mathrm{~kJ} / \mathrm{kg}\)....

Study smarter with the SolutionInn App


