The following is the line-angle drawing of a molecule called silicic acid, an important plant hormone...
Fantastic news! We've Found the answer you've been seeking!
Question:

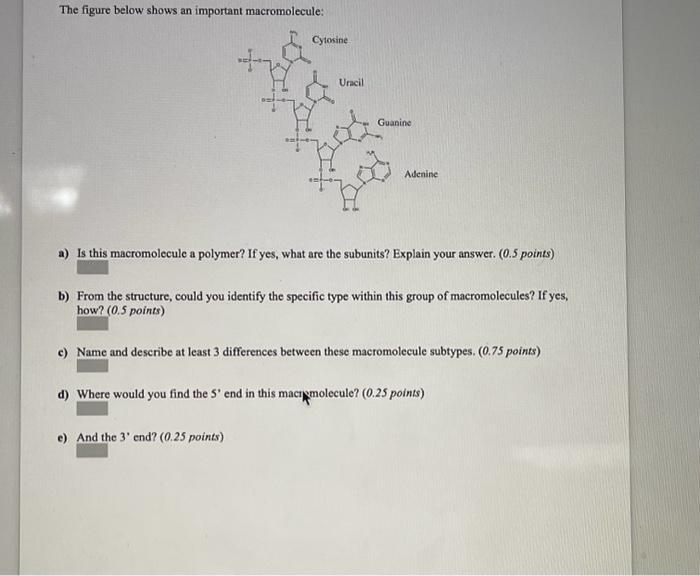
Transcribed Image Text:
The following is the "line-angle" drawing of a molecule called silicic acid, an important plant hormone that has been described to play an essential role in the activation and regulation of multiple responses to biotic and to abiotic. The carbon atoms and the hydrogen atoms bonded to them are not shown but implied. HO HO a) What is the molecular formula for this molecule? (0.25 points each) i) How many H? ii) How many C? iii) How many O? iv) Molecular formula (CxHxOx): b) What are isomers? Use your iv) answer as an example. (0.5 points) c) Can you identify functional groups? (0.33 each) i. Are the functional groups polar or non-polar? ii. What does polar mean in this context? iii. Which functional groups can participate in hydrogen bond formation? d) Why can a C atom form up to four covalent bonds whereas H is able to form only one covalent bond? Explain your answer using your own words. (I point) The figure below shows an important macromolecule: I't Cytosine DIFF Uracil the 3' end? (0.25 points) Guanine IF R Adenine a) Is this macromolecule a polymer? If yes, what are the subunits? Explain your answer. (0.5 points) b) From the structure, could you identify the specific type within this group of macromolecules? If yes, how? (0.5 points) c) Name and describe at least 3 differences between these macromolecule subtypes. (0.75 points) d) Where would you find the 5' end in this macromolecule? (0.25 points) The following is the "line-angle" drawing of a molecule called silicic acid, an important plant hormone that has been described to play an essential role in the activation and regulation of multiple responses to biotic and to abiotic. The carbon atoms and the hydrogen atoms bonded to them are not shown but implied. HO HO a) What is the molecular formula for this molecule? (0.25 points each) i) How many H? ii) How many C? iii) How many O? iv) Molecular formula (CxHxOx): b) What are isomers? Use your iv) answer as an example. (0.5 points) c) Can you identify functional groups? (0.33 each) i. Are the functional groups polar or non-polar? ii. What does polar mean in this context? iii. Which functional groups can participate in hydrogen bond formation? d) Why can a C atom form up to four covalent bonds whereas H is able to form only one covalent bond? Explain your answer using your own words. (I point) The figure below shows an important macromolecule: I't Cytosine DIFF Uracil the 3' end? (0.25 points) Guanine IF R Adenine a) Is this macromolecule a polymer? If yes, what are the subunits? Explain your answer. (0.5 points) b) From the structure, could you identify the specific type within this group of macromolecules? If yes, how? (0.5 points) c) Name and describe at least 3 differences between these macromolecule subtypes. (0.75 points) d) Where would you find the 5' end in this macromolecule? (0.25 points)
Expert Answer:
Answer rating: 100% (QA)
a In The structure of a silicic acid a benzene ring is present along with 2 functional groups OH hydroxyl and COOH carboxylic acid i Number of hydroge... View the full answer

Related Book For 

Operations Management Processes and Supply Chains
ISBN: 978-0133872132
11th edition
Authors: Lee J. Krajewski, Manoj K. Malhotra, Larry P. Ritzman
Posted Date:
Students also viewed these human resource management questions
-
What is the molecular formula for a cyclic alkyne with 14 carbons and two triple bonds?
-
What is the molecular formula for each of the compounds given in Exercise 1.33? (a) (b) (c) (d)
-
What is the molecular formula for each of the following compounds? Consult Figures 1.13 and 1.14 for the abbreviated structural formulas. a. Nicotine b. Adenine c. Limonene d. Coumarin e. Benzene
-
Using the aggregate expenditures table below, answer the questions that follow. a. Compute the APC when income equals $2,300 and the APS when income equals $2,800. b. Compute the MPC and MPS. c. What...
-
In May 2001, unable to get credit from enough of its lenders, housewares retailer Lechters, Inc., filed for Chapter 11 bankruptcy. It then secured new bank financing in the amount of $86 million....
-
Instead of lives-in-being plus 21 years, the rule might be lives-in-being plus 10 years, or lives-in-being plus 35 years. Compare these rules as means for generation-skipping.
-
Hemlock Semiconductor Operations, LLC, and SolarWorld Industries Sachsen GmbH, are both companies involved in the manufacture of components for solar power products. Prior to the lawsuit, the two...
-
Union Bay Plastics is investigating the purchase of automated equipment that would save $100,000 each year in direct labor and inventory carrying costs. This equipment costs $750,000 and is expected...
-
Given that R is the relation defined on TL as follows: "For every m, n = Z, m R n 3|(m-n). (a) Prove that the relation is an equivalence relation. (b) Identify the distinct equivalence classes of...
-
Kaplan Computers manufactures high end computer systems for the graphics design industry. At the present time, the company makes three computers with the following characteristics: Computer 1...
-
In a SHORT CIRCUIT (LOCKED ROTOR) TEST OF THE ASYNCHRONOUS MOTOR V=227V , I= 1.61A , cos(theta) = 0.09 W=36 VA=373 VAR =372 shaft speed=0 Re=39.77ohm Is there any (Pfe) iron loses on the short...
-
-28.1 x 10-6 C 91 Find the net force on 9. +25.5 x 106 C + 92 0.300 m 0.300 m F = force exerted on 92 by 91 F3 = force exerted on 92 by 93 F3 = N [F= F = [?] N Remember: Like charges repel; opposite...
-
A 4900 resistor is connected across a charged 0.850 nF capacitor. The initial current through the resistor, just after the connection is made, is measured to be 0.271 A. Part A What magnitude of...
-
Suppose the student in (Figure 1) is 58kg, and the board being stood on has a 12kg mass. Figure 1.5 m 2.0 m 1 of 1 Part A What is the reading on the left scale? Express your answer in newtons. mleft...
-
Given assemblage of 5 Springs (Figure 1), with stiffness: k = 105 N/m, k2 = 2x105 N/m, k3 = 3x105 N/m, k4 = 4x105 N/m, k5 = 5x105 N/m. These Springs are subjected to a load, P=1000 N at node 4....
-
A thunderstorm drops 1/4 in. of rain on a 50 mi area. The estimated number of fallen raindrops is O 100,000. O 106 O 10% O 107. O 107
-
The production manager at Cape Marine Engines is investigating a new process which makes a 36-inch shaft used in outboard motors. Since this is a new process, she wants to get descriptive statistics...
-
A 2500-lbm car moving at 15 mi/h is accelerated at a constant rate of 15 ft/s 2 up to a speed of 50 mi/h. Calculate force and total time required?
-
Jordanne King, the project manager for Webjets International, Inc., compiled a table showing time estimates for each of the activities of a project to upgrade the company's Web page, including...
-
You are in charge of analyzing five new suppliers of an important raw material and have been given the information shown below (1 = worst, 10 = best). Management has decided that criteria 2 and 3 are...
-
1. How can the management specifically improve the stewarding process at The Phoenician? Using the information provided, create a flowchart illustrating the new process. 2. What are the benefits that...
-
Marfan syndrome is a rare inherited human disorder characterized by unusually long limbs and digits plus defects in the heart (especially the aorta) and the eyes, among other symptoms. Following is a...
-
An individual has the genotype Aa Bb Cc and makes an abnormal gamete with the genotype AaBc. Does this gamete violate the law of independent assortment or the law of segregation (or both)? Explain...
-
A true-breeding tall pea plant was crossed to a true-breeding dwarf plant. What is the probability that an F1 individual will be truebreeding? What is the probability that an F1 individual will be a...

Study smarter with the SolutionInn App


