The following reaction between ethyl alcohol and ethyl bromide forms diethyl ether via a substitution reaction...
Fantastic news! We've Found the answer you've been seeking!
Question:
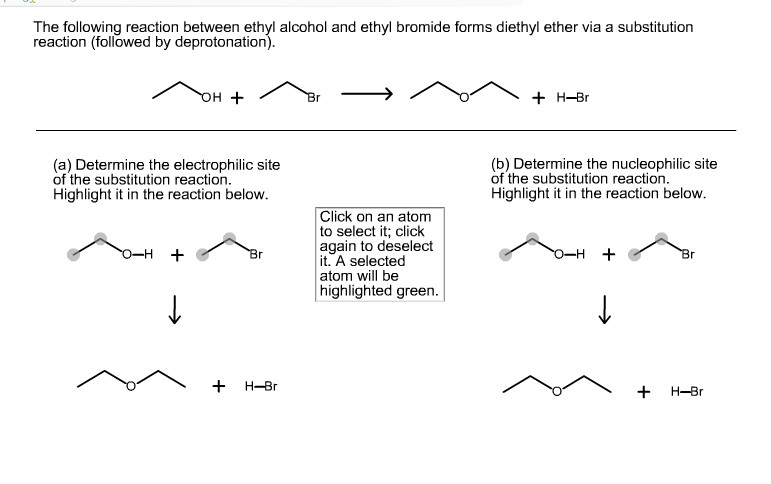
Transcribed Image Text:
The following reaction between ethyl alcohol and ethyl bromide forms diethyl ether via a substitution reaction (followed by deprotonation). (a) Determine the electrophilic site of the substitution reaction. Highlight it in the reaction below. O-H + OH + ↓ + Br H-Br Br Click on an atom to select it; click again to deselect it. A selected atom will be highlighted green. + H-Br (b) Determine the nucleophilic site of the substitution reaction. Highlight it in the reaction below. O-H + ↓ + H-Br The following reaction between ethyl alcohol and ethyl bromide forms diethyl ether via a substitution reaction (followed by deprotonation). (a) Determine the electrophilic site of the substitution reaction. Highlight it in the reaction below. O-H + OH + ↓ + Br H-Br Br Click on an atom to select it; click again to deselect it. A selected atom will be highlighted green. + H-Br (b) Determine the nucleophilic site of the substitution reaction. Highlight it in the reaction below. O-H + ↓ + H-Br
Expert Answer:
Answer rating: 100% (QA)
General Guidance Electrophile is an electron loving and nucleophile is a nucleus loving Electron pai... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Draw and name the product for the following reaction between an alkyne and one equivalent of HCl. Hint/ Explanation: H and Cl? will add to the carbon atoms of the triple bond. The addition is...
-
The reaction between ethyl bromide (C2H5Br) and hydroxide ion in ethyl alcohol at 330 K, C2H5Br(alc) + OH (alc) -- C2H5OH(I) + Br(aIc), is first order each in ethyl bromide and hydroxide ion. When...
-
Consider the following reaction between mercury (II) chloride and oxalate ion: 2 HgCI2(aq) + C2O42- (aq) 2 CIË (aq) + 2 CO2(g) + Hg2CI2(s) The initial rate of this reaction was determined for...
-
A pistoncylinder device contains superheated steam. During an actual adiabatic process, the entropy of the steam will (never, sometimes, always) increase.
-
Abercrombie and Fitch is a leading retailer of casual apparel for men, women, and children. Assume that you are employed as a stock analyst and your boss has just completed a review of the new...
-
Consider a two- player, sequential- move game where each player can choose to play right or left. Player 1 moves first. Player 2 observes player 1s actual move and then decides to move right or left....
-
Stone Brewing Co. is a San Diego brewer that has sold its beers for over two decades. Stone has maintained its trademark and brand from the beginning, registering the STONE mark in 1998. Stone has...
-
(Entries for Retirement and Issuance of Bonds) Friedman Company had bonds outstanding with a maturity value of $500,000. On April 30, 2011, when these bonds had an unamortized discount of $10,000,...
-
You invest $300,000 for new equipment at a manufacturing facility. The equipment is expected to produce $125,000 per year in revenue. The expenses for operating the equipment are $45,000/year. There...
-
On January 1, 2023, Psalm and Trisha formed PS Partnership. The articles of co-partnership provides that profit or loss shall be distributed accordingly: 15% interest on average capital balance....
-
Cause-and-effect diagrams are also known as A. Pareto charts. B. Target specification graphs. C. Fsh-bone charts. D. Flowcharts.
-
You raise an excellent point; this should be researched and selected accordingly when providing tax breaks to industries, sectors, or groups. However, in our society, whenever someone or a group...
-
1. Stevie needs rug money. Stevie plays the California lottery and wins $1 million to be paid out at $100,000 per year (at the end of each year) for the next 10 years. Current interest rates on high...
-
Before there were no taxes on towels. The price of a towel was $8 and the quantity purchased was 300 towels. Then the government imposed a tax of $2 per towel. As a result, the price of towels for...
-
Cigarettes have been used as money in exceptional circumstances. What is the most likely reason why they are not acceptable as money under normal circumstances?
-
Suppose Kenji, an economist at a business school in Georgia, and Lucia, an economist at a public television show, are arguing about health insurance.The following dialogue shows an excerpt of their...
-
Let ACBCR, and assume that B is bounded. i) Prove that A is bounded ii) Prove rigorously that inf(B) inf(A) sup(A) sup(B). Extrelse 5. Find sets A, B CR such that x < y for all x EA and y E B, and...
-
Write a paper by answer the following question: Should Recycling Be Mandatory?
-
In the problem-solving feature above, methylcyclohexanone was seen to react at its unsubstituted carbon. Try to write a mechanism for the same reaction at the methyl-substituted carbon atom, and...
-
The standard 13C NMR spectrum of phenyl propanoate is shown here. Predict the appearance of the DEPT-90 and DEPT-135 spectra. 13C NMR 0-C-CH2CH3 pheny! propanoate 200 180 160140 10 100 80 40 20 0 8...
-
Predict the products of the following reactions. (a) Cyclopentanone + Br2 in acetic acid (b) 1-phenylethanol + excess I2 in base (c) (d) (e) (f) (g) (h) (i) CH (1) LDA CH CH,CH,CH,Br Ph TOH Ph (2)...
-
A candlestick chart is similar to a bar chart except that the candlestick chart: A. Represents upward movements in price with Xs. B. Also graphically shows the range of the periods highs and lows. C....
-
What considerations might encourage a market analyst to rely more on a top-down or bottom-up forecast of S&P 500 operating earnings?
-
Which of the following advantages listed by Ryan with respect to the earnings-based approach studied by Atkinson is most likely correct? The model A. Can be used for non-U.S. equity markets. B....

Study smarter with the SolutionInn App


