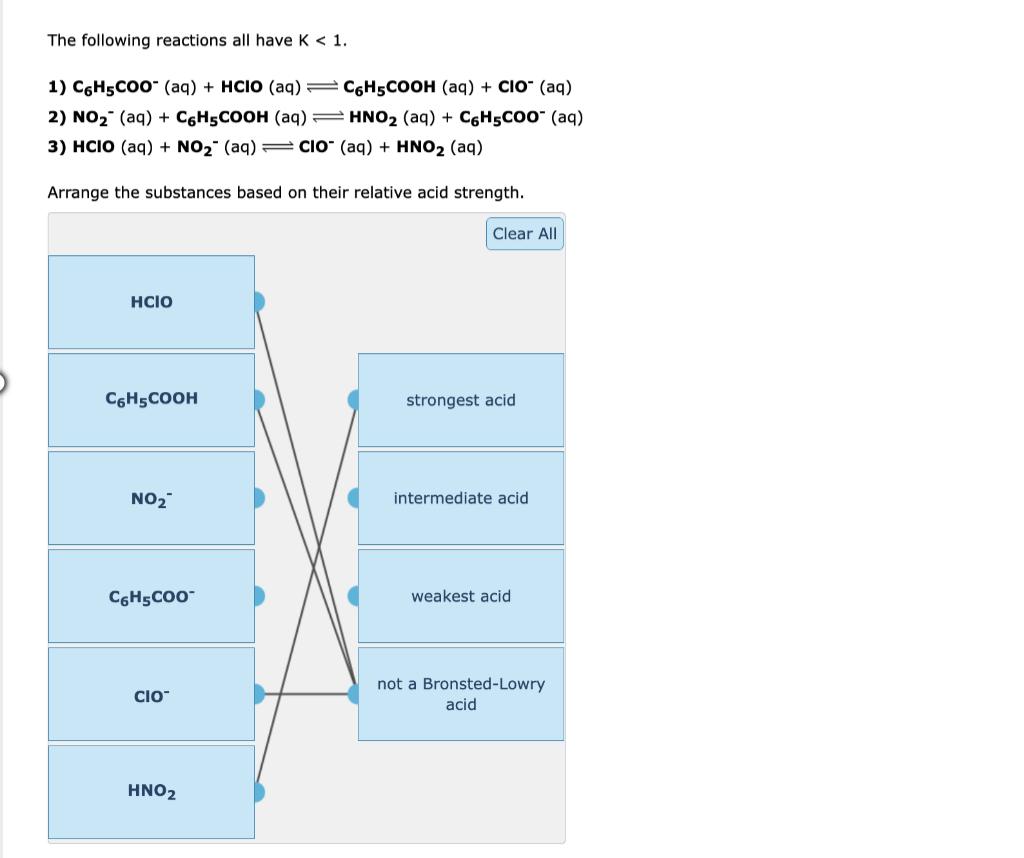
The following reactions all have K < 1. 1) C6H5COO (aq) + HCIO (aq) = C6H5COOH...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
The following reactions all have K < 1. 1) C6H5COO (aq) + HCIO (aq) = C6H5COOH (aq) + CIO- (aq) 2) NO₂ (aq) + C6H5COOH (aq) HNO₂ (aq) + C6H5COo- (aq) 3) HCIO (aq) + NO₂ (aq) CIO (aq) + HNO2 (aq) Arrange the substances based on their relative acid strength. Clear All HCIO C6H5COOH strongest acid NO₂ intermediate acid C6H5COO™ weakest acid CIO™ not a Bronsted-Lowry acid HNO2 The following reactions all have K < 1. 1) C6H5COO (aq) + HCIO (aq) = C6H5COOH (aq) + CIO- (aq) 2) NO₂ (aq) + C6H5COOH (aq) HNO₂ (aq) + C6H5COo- (aq) 3) HCIO (aq) + NO₂ (aq) CIO (aq) + HNO2 (aq) Arrange the substances based on their relative acid strength. Clear All HCIO C6H5COOH strongest acid NO₂ intermediate acid C6H5COO™ weakest acid CIO™ not a Bronsted-Lowry acid HNO2
Expert Answer:
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Posted Date:
Students also viewed these chemistry questions
-
Arrange the following substances in order of increasing melting point. Si H-C-O-H H-C-C-H NaCl
-
Arrange the following substances (1 mole each) in order of increasing entropy at 25C: (a) Ne(g), (b) SO2(g), (c) Na(s), (d) NaCl(s), (e) H2(g). Give the reasons for your arrangement.
-
Arrange six departments into a 2 3 floor grid so that these conditions are satisfied: 1 is close to 2:5 is close to 2,5, and 6: and 3 is not close to 1 or 2.
-
The 2017 financial statements for the Thor and Gunnar companies are summarized below. These two companies are in the same business and in the same province but in different cities. One-half of Thor?s...
-
Surgical Supplies Corporation paid a dividend of $1.12 per share over the last 12 months. The dividend is expected to grow at a rate of 25 percent over the next three years (supernormal growth). It...
-
Which of the series converge absolutely, which converge, and which diverge? Give reasons for your answers. 00 n=1 (-1)n-1 n? + 2n + 1 2
-
Find the dynamic response of the stepped bar shown in Fig. 12.37 (a) when its free end is subjected to the load given in Fig. 12.37(b). P(t) Arca - 4A Area A Po P(1) (a) FIGURE 12.37 Stepped bar with...
-
Osage Corporation issued 2,000 shares of stock. Instructions Prepare the entry for the issuance under the following assumptions. (a) The stock had a par value of $5 per share and was issued for a...
-
create an erd diagram for student registration form and daATABASE CODE TO CREATE THE TABLE...I will tip if this work is done in less than 3 hours
-
At June 30, 2022, the end of its most recent fiscal year, Green River Computer Consultants post-closing trial balance was as follows: The company underwent a major expansion in July. New staff was...
-
Evaluate the corporate structure of EarthWear Clothiers and their annual report and then summarize your findings. Evaluate the following for your auditing firm, Willis & Adams: independence,...
-
An advantage when one corporation controls another is that the controlling entity's management has the ability to transfer resources between the two legal entities as needed. For example, the...
-
Preparing a Profit Equation to Predict Future Profits Picnic Time produces a picnic basket that is sold for $ 1 2 0 per unit. Assume the company produced and sold 4 , 0 0 0 baskets during July. There...
-
Analyse the security procedures that BMW must implement to guarantee that inventory is recorded accurately with as few errors as feasible.
-
ACh is the only one labeled correctly. Identify/label the rest. ganglion NE postsynaptic neuron ganglion postsynaptic neuron nicotinic receptors NE ACh ACh presynaptic neuron Reset Zoom nicotinic...
-
What is the value of the z variable after this Java code executes? int x = 77; int y=88; int z = 99; x = y; z = y;
-
Why are Accounts Receivable opening balances not posted to the General Ledger? a. Accounts Receivable is a control account. b. The Accounts Receivable amount is already in the Accounts Receivable...
-
The Taylor's series expansion for cosx about x = 0 is given by: where x is in radians. Write a user-defined function that determines cosx using Taylor's series expansion. For function name and...
-
At a certain temperature, K = 9.1 10 -4 for the reaction Calculate the concentrations of Fe 3+ , SCN - , and FeSCN 2+ in a solution that is initially 2.0 M FeSCN 2+ . 3+ FeSCN2+ (aq) Fe+ (aq) + SCN-...
-
Calculate the equilibrium concentrations of NH 3 , Cu 2+ , Cu(NH 3 ) 2+ , Cu(NH 3 ) 2 2+ , Cu(NH 3 ) 3 2+ , and Cu(NH 3 ) 4 2+ in a solution prepared by mixing 500.0 mL of 3.00 M NH 3 with 500.0 mL...
-
Estradiol is a female hormone with the following structure: How many chiral carbon atoms are in estradiol? HO CH3 OH
-
For the conditions of Sample Problem 5/17, determine the angular acceleration of \(A C\) and the acceleration of \(A\) relative to the rotating slot in \(\operatorname{arm} O D\). Problem 5/17, The...
-
Aircraft \(B\) has a constant speed of \(150 \mathrm{~m} / \mathrm{s}\) as it passes the bottom of a circular loop of 400-m radius. Aircraft \(A\) flying horizontally in the plane of the loop passes...
-
Examples of random variables used in this chapter included (a) the sex of the next person you meet, (b) the number of times a wireless network fails, (c) the time it takes to commute to school, (d)...

Study smarter with the SolutionInn App



