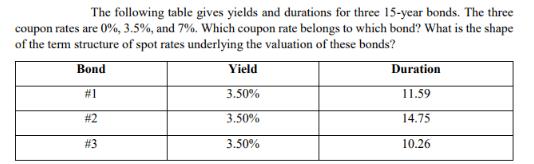
The following table gives yields and durations for three 15-year bonds. The three coupon rates are...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
The following table gives yields and durations for three 15-year bonds. The three coupon rates are 0%, 3.5%, and 7%. Which coupon rate belongs to which bond? What is the shape of the term structure of spot rates underlying the valuation of these bonds? Bond Yield #1 3.50% 3.50% 3.50% # 2 # 3 Duration 11.59 14.75 10.26 The following table gives yields and durations for three 15-year bonds. The three coupon rates are 0%, 3.5%, and 7%. Which coupon rate belongs to which bond? What is the shape of the term structure of spot rates underlying the valuation of these bonds? Bond Yield #1 3.50% 3.50% 3.50% # 2 # 3 Duration 11.59 14.75 10.26
Expert Answer:
Answer rating: 100% (QA)
To determine which coupon rate belongs to which bond we need to match the given yields and durations ... View the full answer

Related Book For 

Posted Date:
Students also viewed these finance questions
-
The following table gives the systolic blood pressure (SBP), body size (QUET), age (AGE), and smoking history (SMK = 0 if a nonsmoker, SMK = 1 if a current or previous smoker) for a hypothetical...
-
The following table gives the prices of bonds *Half the stated coupon is paid every six months a) Calculate zero rates for maturities of 6 months, 12 months, 18 months, and 24 months. b) What are the...
-
The United Students Against Sweatshops (USAS) was formed by students on U.S. and Canadian college campuses in 1999 to protest labor exploitation in the apparel industry. Clark University sociologist...
-
8 D D D D P P P P P P P P P P P 10 Functions What is defusion. What is osnosis What is Tornicity The power house of the cell functions of the plasma? What is facilitated defusion? do they need ATP?...
-
Multiple Choice Questions 1. A manufacturing company is considering purchasing a new machine that doubles capacity from 500 to 1,000 units per week. The machine will occupy approximately 500 square...
-
In the event of an explosive combustion of vapor at atmospheric pressure, the vapor cloud can be modeled as adiabatic because the combustion occurs so rapidly. The vapor cloud expands rapidly due to...
-
Amanda Forbes was hired as a nail technician by Showmann, Inc., in 2011. In 2017, Forbes attended a work-related holiday party where Showmann distributed raffle tickets to employees. One of the...
-
Mile-High Foods, Inc., was formed in March 2011 to provide prepackaged snack boxes for a new low cost regional airline beginning on April 1. The company has just leased warehouse space central to the...
-
Fickle Sickles collects 15,000 checks per 365-day year with average amount $170 and total delay 5 days. A lockbox system would reduce that delay to 3 days, and it would also reduce FISI's check...
-
Casey Morgan is a single taxpayer, social security number 412-34-5670, who lives at 582 Brockton Lane, Columbus, OH 43081. Casey has income from a job as a manager, interest and dividend income, and...
-
Write a detailed paper on Industrial (B2B) Sales Management about Boeing Corporation
-
The following is the residual momentum observer derivation. Robot dynamics is given as follows Define momentum: p = M(q)q M + Cq + g = t+r Compute p using t, Text, g, C, and q
-
Give the exact parent peak mass of the compound C6H6C16. Assume that ONLY the most common isotope is present. Give the mass to 4 decimal places. The masses (in Daltons) of selected isotopes and their...
-
Briefly describe what rethinking the employee experience of remote workers is, along with how and why it will have the most significant impact on the workplace. Then, explain how it could be applied...
-
An ideal monatomic gas expands isothermally from 0.550 m to 1.25 m at a constant temperature of 760 K. If the initial pressure is 1.11 x 105 Pa find the following. (a) the work done on the gas -54400...
-
1. Use a stack to determine if the expression's parentheses are balanced. develop a stack trace like in our lecture (and page 360) that shows each step using a series of boxes....
-
26. What is the pH of a 0.1M solution of ascorbic acid? Forascorbic acid, pKa1 = 4.10, pKa2 = 11.79. a. 1.05 b. 2.55 c. 2.76 d. 3.58 e. 4.40 27. b is the answer 27) In a solution buffered by 40 mM...
-
Four GWU students have been selected to taste food sold by 3 different food trucks labeled as food truck A, B and C on H & 22nd Streets every Monday for 3-weeks. For each student, food trucks are...
-
The price of a 90-day Treasury bill is quoted as 10.00. What continuously compounded return (on an actual/365 basis) does an investor earn on the Treasury bill for the 90-day period?
-
Show that the BlackScholesMerton formula for a call option gives a price that tends to max (So- K,0)
-
It is May 5, 2013. The quoted price of a government bond with a 12% coupon that matures on July 27, 2024, is 110-17. What is the cash price?
-
What is an off-by-one error?
-
Will the universality of management continue to be true in the future? Why or why not?
-
Describe what a manager does. How does the work of managers differ from that of nonmanagerial employees?

Study smarter with the SolutionInn App


