The free energy of a gas. N particles in a volume V and at a temperature...
Fantastic news! We've Found the answer you've been seeking!
Question:
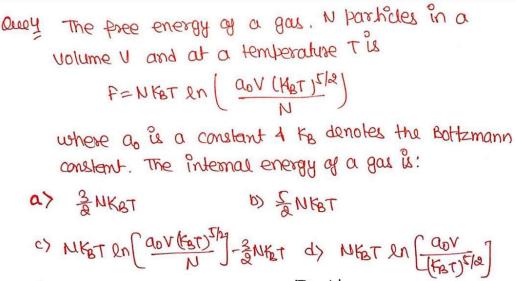
Transcribed Image Text:
The free energy of a gas. N particles in a volume V and at a temperature T is F=NKBT ln ( aoV (KbT)^5/2 /N) where a is a constant of Kb denotes the bottzmann constant. The internal energy of a gas is: The free energy of a gas. N particles in a volume V and at a temperature T is F=NKBT ln ( aoV (KbT)^5/2 /N) where a is a constant of Kb denotes the bottzmann constant. The internal energy of a gas is:
Expert Answer:
Related Book For 

Fundamentals of biochemistry Life at the Molecular Level
ISBN: 978-0470547847
4th edition
Authors: Donald Voet, Judith G. Voet, Charlotte W. Pratt
Posted Date:
Students also viewed these physics questions
-
Gruneisen constant (a) Show that the free energy of a phonon mode of frequency w is k B T in [2sinh (hw/2k B T)]. It is necessary to retain the zero-point energy hw to obtain this result. (b) If is...
-
The average kinetic energy of a gas molecule can be found from which of these quantities? (a) Pressure only (b) Number of molecules only (c) Temperature only (d) Pressure and temperature are both...
-
The free energy of formation of one mole of compound refers to a particular chemical equation. For each of the following, write that equation. a. KBr(s) b. CH3Cl(l) c. H2S(g) d. AsH3(g)
-
Nevada Department Stores is planning to sell its Spring Valley. Fernley, and Winchester stores. The firm expects to sell each of the three stores for the same, positive cash flow of SD. The firm...
-
The heat flux q is the quantity of heat flowing through a unit area of a material per unit time. It can be computed with Fouriers law, Where J has units of J/m 2 /s or W/m 2 and k is a coefficient of...
-
What are two ways in which Madison believes factions can be dealt with?
-
Upland Metals Company had 20,000,000 shares of \(\$ 0.01\) par value common stock outstanding which had been sold for an aggregate amount of \(\$ 300,000,000\). The company's shares are traded on the...
-
When real estate agents sell their own, rather than clients, houses, they leave the houses on the market for a longer time (10 days longer on average) and wind up with better prices (2% higher on...
-
You live in an area that has a possibility of incurring a massive earthquake, so you are considering buying earthquake insurance on your home at an annual cost of $180. The probability of an...
-
You want to understand if the final model can be improved by selecting customers whose properties are in either Urban or Semi-urban area. Which of the following model did you find from your analysis...
-
A vaccine is being purified from 20 L of cell culture supernatant by adsorption. The adsorption process follows Langmuir isotherm KL = 0.5 g/L and CBmax = 1.5 g/g. Theconcentration of the vaccine in...
-
Cintu is a large marketer and distributor of food service products serving restaurants, hotels, schools, hospitals, and other institutions. The following transactions are typical of those that...
-
The marketing strategies of a business must be understood in order to differentiate business-to-business (B2B) from business-to-consumer (B2C). Business-to- business marketing is a form of marketing...
-
A number of lawsuits have been filed against pharmaceutical companies in the U.S. alleging negligence, strict liability, and even intentional harm. Many current lawsuits involve the manufacture,...
-
Recall that two angles are complements of each other if their sum is 90. Angle A and angle B are complementary angles and angle A is 22 more than three times angle B. Find the measures of angle A and...
-
How can we incorporate personas into our products and brands? 2. Content writing plays a very important role in our digital platforms. In what ways does content convert into sales? Let us know what...
-
1) Perform Sampling with intervals of 3 seconds. 10 9 27 18 36 10 T 2) How many bits are required to present a range from 1 to 33? 3) Describe how Claire would send a message to David using...
-
The overall reaction and equilibrium constant value for a hydrogenoxygen fuel cell at 298 K is 2H 2 (g) + O 2 (g) 2H 2 O(l) K = 1.28 10 83 a. Calculate E cell and G 8 at 298 K for the fuel cell...
-
Consider the mitochondrial ADP-ATP translocator and the Pi-H+ symport protein. How do the activities of the two transporters affect the electrochemical gradient across the mitochondrial membrane?
-
Compare the molecular mass (M) of horse heart apomyoglobin calculated from the two adjacent peaks at 1884.7 and 1696.3, and 893.3 and 848.7 (Figure 5-16b). How do these results compare with those in...
-
Calculate the pH of a 200 mL solution of pure water to which has been added 50 mL of 1 mM HCl.
-
Consider estimation of the tail probability \(\mu=\mathbb{P}[X \geqslant \gamma]\) of some random variable \(X\), where \(\gamma\) is large. The crude Monte Carlo estimator of \(\mu\) is \[...
-
Let \(X\) and \(Y\) be random variables (not necessarily independent) and suppose we wish to estimate the expected difference \(\mu=\mathbb{E}[X-Y]=\mathbb{E} X-\mathbb{E} Y\). (a) Show that if \(X\)...
-
One of the test cases in [70] involves the minimization of the Hougen function. Implement a cross-entropy and a simulated annealing algorithm to carry out this optimization task.

Study smarter with the SolutionInn App



