The gas in Question 1 goes through a choke at the surface with free water present...
Fantastic news! We've Found the answer you've been seeking!
Question:


Transcribed Image Text:
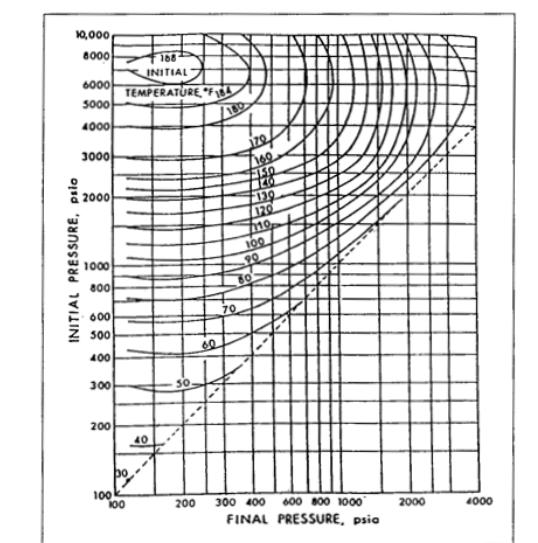
The gas in Question 1 goes through a choke at the surface with free water present under following conditions. Upstream of the choke : P = 2500 psi 100 F T= (i) At 100 F determine the lowest pressure this gas can be expanded to across the choke without hydrate formation (ii) At 2000 psi what is the lowest temperature in the flow line before hydrates form Use data provided at the end. Please explain how you calculate your answers MW of Air = 28.97 Ib/lb mole 10,000 8000 168 INITIAL 6000 TEMPERATURE "FL 5000 4000 3000 FI50 2000 120 100 90 1000 800 60 600 70 500 60 400 300 200 40 100 100 200 300 400 600 800 1000 2000 4000 FINAL PRESSURE, psia INITIAL PRESSURE, psio 0.065 0.06 0.055 0.05 0.045 0.04 0.035 0.03 0.025 0.02 0.015 0.01 Pressure (psia) 00001 0006 -0008 Viscos ity (cp) 700 F 9000 Enthalpy-entropy diagram + for 0.7 gravity natural gas 8000 600, 7000- pPc à 667 psia, pTc = 392° R 500 6000 500 5000 400. 4000 300 3000 200 2000 100 1000 200 32 F 100 - 1000 -2000 8 10 12 -14 -12 -10 -8 -6 -4 -2 0 2 4 Entropy, Btu per pound-mole per degree 0002 os 5000 ersd 000'01 000i pool 300 Enthalpy, Btu per pound-mole The gas in Question 1 goes through a choke at the surface with free water present under following conditions. Upstream of the choke : P = 2500 psi 100 F T= (i) At 100 F determine the lowest pressure this gas can be expanded to across the choke without hydrate formation (ii) At 2000 psi what is the lowest temperature in the flow line before hydrates form Use data provided at the end. Please explain how you calculate your answers MW of Air = 28.97 Ib/lb mole 10,000 8000 168 INITIAL 6000 TEMPERATURE "FL 5000 4000 3000 FI50 2000 120 100 90 1000 800 60 600 70 500 60 400 300 200 40 100 100 200 300 400 600 800 1000 2000 4000 FINAL PRESSURE, psia INITIAL PRESSURE, psio 0.065 0.06 0.055 0.05 0.045 0.04 0.035 0.03 0.025 0.02 0.015 0.01 Pressure (psia) 00001 0006 -0008 Viscos ity (cp) 700 F 9000 Enthalpy-entropy diagram + for 0.7 gravity natural gas 8000 600, 7000- pPc à 667 psia, pTc = 392° R 500 6000 500 5000 400. 4000 300 3000 200 2000 100 1000 200 32 F 100 - 1000 -2000 8 10 12 -14 -12 -10 -8 -6 -4 -2 0 2 4 Entropy, Btu per pound-mole per degree 0002 os 5000 ersd 000'01 000i pool 300 Enthalpy, Btu per pound-mole The gas in Question 1 goes through a choke at the surface with free water present under following conditions. Upstream of the choke : P = 2500 psi 100 F T= (i) At 100 F determine the lowest pressure this gas can be expanded to across the choke without hydrate formation (ii) At 2000 psi what is the lowest temperature in the flow line before hydrates form Use data provided at the end. Please explain how you calculate your answers MW of Air = 28.97 Ib/lb mole 10,000 8000 168 INITIAL 6000 TEMPERATURE "FL 5000 4000 3000 FI50 2000 120 100 90 1000 800 60 600 70 500 60 400 300 200 40 100 100 200 300 400 600 800 1000 2000 4000 FINAL PRESSURE, psia INITIAL PRESSURE, psio 0.065 0.06 0.055 0.05 0.045 0.04 0.035 0.03 0.025 0.02 0.015 0.01 Pressure (psia) 00001 0006 -0008 Viscos ity (cp) 700 F 9000 Enthalpy-entropy diagram + for 0.7 gravity natural gas 8000 600, 7000- pPc à 667 psia, pTc = 392° R 500 6000 500 5000 400. 4000 300 3000 200 2000 100 1000 200 32 F 100 - 1000 -2000 8 10 12 -14 -12 -10 -8 -6 -4 -2 0 2 4 Entropy, Btu per pound-mole per degree 0002 os 5000 ersd 000'01 000i pool 300 Enthalpy, Btu per pound-mole The gas in Question 1 goes through a choke at the surface with free water present under following conditions. Upstream of the choke : P = 2500 psi 100 F T= (i) At 100 F determine the lowest pressure this gas can be expanded to across the choke without hydrate formation (ii) At 2000 psi what is the lowest temperature in the flow line before hydrates form Use data provided at the end. Please explain how you calculate your answers MW of Air = 28.97 Ib/lb mole 10,000 8000 168 INITIAL 6000 TEMPERATURE "FL 5000 4000 3000 FI50 2000 120 100 90 1000 800 60 600 70 500 60 400 300 200 40 100 100 200 300 400 600 800 1000 2000 4000 FINAL PRESSURE, psia INITIAL PRESSURE, psio 0.065 0.06 0.055 0.05 0.045 0.04 0.035 0.03 0.025 0.02 0.015 0.01 Pressure (psia) 00001 0006 -0008 Viscos ity (cp) 700 F 9000 Enthalpy-entropy diagram + for 0.7 gravity natural gas 8000 600, 7000- pPc à 667 psia, pTc = 392° R 500 6000 500 5000 400. 4000 300 3000 200 2000 100 1000 200 32 F 100 - 1000 -2000 8 10 12 -14 -12 -10 -8 -6 -4 -2 0 2 4 Entropy, Btu per pound-mole per degree 0002 os 5000 ersd 000'01 000i pool 300 Enthalpy, Btu per pound-mole The gas in Question 1 goes through a choke at the surface with free water present under following conditions. Upstream of the choke : P = 2500 psi 100 F T= (i) At 100 F determine the lowest pressure this gas can be expanded to across the choke without hydrate formation (ii) At 2000 psi what is the lowest temperature in the flow line before hydrates form Use data provided at the end. Please explain how you calculate your answers MW of Air = 28.97 Ib/lb mole 10,000 8000 168 INITIAL 6000 TEMPERATURE "FL 5000 4000 3000 FI50 2000 120 100 90 1000 800 60 600 70 500 60 400 300 200 40 100 100 200 300 400 600 800 1000 2000 4000 FINAL PRESSURE, psia INITIAL PRESSURE, psio 0.065 0.06 0.055 0.05 0.045 0.04 0.035 0.03 0.025 0.02 0.015 0.01 Pressure (psia) 00001 0006 -0008 Viscos ity (cp) 700 F 9000 Enthalpy-entropy diagram + for 0.7 gravity natural gas 8000 600, 7000- pPc à 667 psia, pTc = 392° R 500 6000 500 5000 400. 4000 300 3000 200 2000 100 1000 200 32 F 100 - 1000 -2000 8 10 12 -14 -12 -10 -8 -6 -4 -2 0 2 4 Entropy, Btu per pound-mole per degree 0002 os 5000 ersd 000'01 000i pool 300 Enthalpy, Btu per pound-mole The gas in Question 1 goes through a choke at the surface with free water present under following conditions. Upstream of the choke : P = 2500 psi 100 F T= (i) At 100 F determine the lowest pressure this gas can be expanded to across the choke without hydrate formation (ii) At 2000 psi what is the lowest temperature in the flow line before hydrates form Use data provided at the end. Please explain how you calculate your answers MW of Air = 28.97 Ib/lb mole 10,000 8000 168 INITIAL 6000 TEMPERATURE "FL 5000 4000 3000 FI50 2000 120 100 90 1000 800 60 600 70 500 60 400 300 200 40 100 100 200 300 400 600 800 1000 2000 4000 FINAL PRESSURE, psia INITIAL PRESSURE, psio 0.065 0.06 0.055 0.05 0.045 0.04 0.035 0.03 0.025 0.02 0.015 0.01 Pressure (psia) 00001 0006 -0008 Viscos ity (cp) 700 F 9000 Enthalpy-entropy diagram + for 0.7 gravity natural gas 8000 600, 7000- pPc à 667 psia, pTc = 392° R 500 6000 500 5000 400. 4000 300 3000 200 2000 100 1000 200 32 F 100 - 1000 -2000 8 10 12 -14 -12 -10 -8 -6 -4 -2 0 2 4 Entropy, Btu per pound-mole per degree 0002 os 5000 ersd 000'01 000i pool 300 Enthalpy, Btu per pound-mole
Expert Answer:
Answer rating: 100% (QA)
93 we have given statements a All ten runners finished ... View the full answer

Posted Date:
Students also viewed these physics questions
-
Please explain how you will be utilizing technology to make your business more effective and efficient. Explore the cloud technology concept. Discuss the importance of information technology planning...
-
Air flows at Ma = 3 and p = 10 psia toward a wedge of 16° angle at zero incidence, as in Fig. P9.132 (a) If the pointed edge is forward, what is the pressure at point A? If the blunt edge is...
-
A 1.00-mol sample of an ideal monatomic gas is at an initial temperature of 300 K. The gas undergoes an is volumetric process acquiring 500 J of energy by heat. It then undergoes an isobaric process...
-
A circular transistor of 10-mm diameter is cooled by impingement of an air jet exiting a 2-mm-diameter round nozzle with a velocity of 20 m/s and a temperature of 15C. The jet exit and the exposed...
-
Why is user acceptance important?
-
The following items are from the 2006 balance sheet of Kellogg Company. (All dollars are in millions.) Common stock ............$ 396.9 Other assets ............5,471.4 Notes payablecurrent...
-
Task: Use Excel and the Solver add-in to explore the effect of various resource constraints on the optimal product mix. a. Read the article Boost Profits With Excel, by James A. Weisel in the...
-
Identify the accounting concept or principle that gives the most direction on how to account for each of the following situations: a. Salary expense of $38,000 is accrued at the end of the period to...
-
Assume that Conner Co. negotiated a forward contract to purchase 2,000,000 British pounds in 60 days. The 60 day forward rate was $1.40 per British pound. The pounds to be purchased were to be used...
-
Find each function f(x) given, (a) find any three ordered pair solutions (a,b), then (b) algebraically compute f(x), and (c) verify the ordered pairs (a,b) satisfy f(x). f(x)=x-2 f(x)=x +3 f(x) = x +...
-
Wishes is a nonprofit company that specializes in selling items online that are donated by celebrities, such as clothing, jewelry, purses, and many other forms of memorabilia. The profits from Wishes...
-
One side of tank contains an ideal gas at 560 C and the other side is evacuated. The evacuated side has the 6 times volume of the side filled with gas. The initial specific volume of the ideal gas is...
-
n D 16 : Use the product code breakdown provided at the top of the worksheet as a guide to create appropriate nested functions to complete the following: If the product is Premium insert the product...
-
Assuming the current stock price for Apple is $160, the expected continuously com- pounded annual growth rate for Apple is AAPL = 0.05, and the volatility AAPL is such that AAPL 0.1, what is the...
-
Find 5 x5 5 (+2x+4) dz da +2x+4 dx = +4)dx= Question Help: Video Written Example + C
-
Please find the following derivative a) f(t)=sin(t)
-
[The following information applies to the questions displayed below.] On January 1, 2024, Twister Enterprises, a manufacturer of a variety of transportable spin rides, issues $420,000 of 6% bonds,...
-
Why do CPA firms sometimes use a combination of positive and negative confirmations on the same audit?
-
Why would intangible assets create significant difficulties in international transfer pricing?
-
Go to the Ernst \& Young Web site (https://www. ey.com/Publication/vwLUAssets/ey-worldwide-transfer-pricing-reference-guide-2018-19/\$FILE ley-worldwide-transfer-pricing-reference-guide-2018-19.pdf)...
-
In Example 15-2, operation is at a pseudo-steady state. Brainstorm alternative designs for this diffusion measurement. Example 15-2 Pure ethanol is contained at the bottom of a long, vertical tube...

Study smarter with the SolutionInn App

