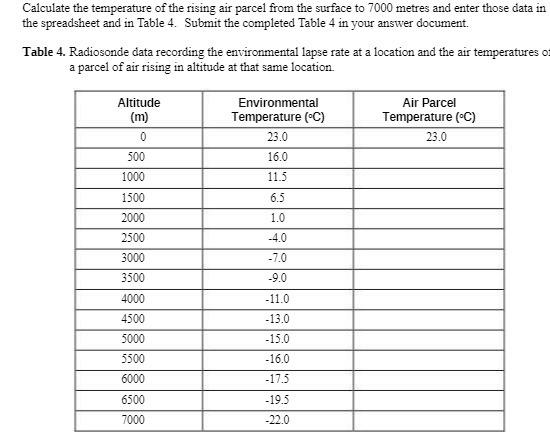
Calculate the temperature of the rising air parcel from the surface to 7000 metres and enter...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Calculate the temperature of the rising air parcel from the surface to 7000 metres and enter those data in the spreadsheet and in Table 4. Submit the completed Table 4 in your answer document. Table 4.Radiosonde data recording the environmental lapse rate at a location and the air temperatures of a parcel of air rising in altitude at that same location. Altitude (m) 0 500 1000 1500 2000 2500 3000 3500 4000 4500 5000 5500 6000 6500 7000 Environmental Temperature (°C) 23.0 16.0 11.5 6.5 1.0 -4.0 -7.0 -9.0 -11.0 -13.0 -15.0 -16.0 -17.5 -19.5 -22.0 Air Parcel Temperature (°C) 23.0 Calculate the temperature of the rising air parcel from the surface to 7000 metres and enter those data in the spreadsheet and in Table 4. Submit the completed Table 4 in your answer document. Table 4.Radiosonde data recording the environmental lapse rate at a location and the air temperatures of a parcel of air rising in altitude at that same location. Altitude (m) 0 500 1000 1500 2000 2500 3000 3500 4000 4500 5000 5500 6000 6500 7000 Environmental Temperature (°C) 23.0 16.0 11.5 6.5 1.0 -4.0 -7.0 -9.0 -11.0 -13.0 -15.0 -16.0 -17.5 -19.5 -22.0 Air Parcel Temperature (°C) 23.0
Expert Answer:
Answer rating: 100% (QA)
To calculate the temperature of the rising air parcel we will assume a dry adiabatic lapse rate of 9... View the full answer

Related Book For 

Posted Date:
Students also viewed these mathematics questions
-
For low-speed (nearly incompressible) gas flow, the stagnation pressure can be computed from Bernoullis equation (a) For higher subsonic speeds, show that the isentropic relation (9.28a) can be...
-
A nonuniform, but spherically symmetric, distribution of charge has a charge density p (r) given as follows: Where P0 = 3Q/πR3 is a positive constant. (a) Show that the total charge contained in...
-
A smooth vertical tube having two different sections is open from both ends and equipped with two pistons of different areas (Fig. 2.i). Each piston slides within a respective tube section. One mole...
-
On October 1, 2024, Modern Company purchased a patent for $153,600 cash. Although the patent gives legal protection for 20 years, the patent is expected to be used for only eight years. Requirements...
-
When purchasing a product on the Web, how important is the visibility of your supply chain for this product? Why? Does the importance differ for different products?
-
You can use a simple technique to find the center of gravity of an irregular shape. Figure Q7. 10 shows a cardboard cutout of the outline of the continental United States. The map is suspended from...
-
The following scenarios describe situations faced by hypothetical companies that currently have a centralized organization structure. As you review each of the scenarios, provide your opinion as to...
-
Spencer Company manufactures and sells three products. Relevant per unit data concerning each product are given below. Instructions (a) Compute the contribution margin per unit of the limited...
-
Imagine that your friend Mary has normal metabolism, but that her brother John has galactosemia. Mary and John's parents, Patricia and Les, are unaffected, although their maternal grandmother, Jean,...
-
A spring with an unstrained length of 0.074 m and a spring constant of 2.4 N/m hangs vertically downward from the ceiling. A uniform electric field directed vertically upward fills the region...
-
Consider the list of integers 12 32 22 15 43 to be inserted into a hash table of size 11 using the hash function h(key)= key % (TABLESIZE). a) Assume linear probing for insertion into the hash table....
-
A certain reaction has an activation energy of 62.0 kJ/mol and a frequency factor of A = 3.10x102 M-1.s. What is the rate constant, k, of this reaction at 29.0 C ? Express your answer with the...
-
One of the following structures does not fit with the others. Circle it. Justify your choice.coOm. U x F
-
Convert henry's constant K _ h from 3 * 1 0 ^ 3 M / atm to henry's constant K _ h in atm * m ^ 3 / mol units
-
Consider a 169 lb individual running at about 18.3 miles per hour. What is the de Broglie wavelength of this runner? Chint: consider your units, and the conversions that may be necessary: Planks...
-
Consider two food dyes one is red and one is blue. a ) Which has a smaller HOMO - LUMO gap? b ) Will this absorb at a higher or lower wavelength? Green Blue Yellow primary primary complementary...
-
you're considering applying for a $2,000 loan that will be repaid with equal year-end payments over the next 13 years. If the annual interest rate of the loan is 6%, how much would you pay?
-
According during to the IRS, individuals filing federal income tax returns prior to March 31 received an average refund of $1,088 in 2018. Consider the population of "last-minute" filers who mail...
-
For the following exercises, use the formula for the sum of the first n terms of each arithmetic sequence. 3.2 + 3.4 + 3.6 + + 5.6
-
For the following exercises, find the decomposition of the partial fraction for the irreducible non repeating quadratic factor. x + 2x + 40 x - 125
-
For the following exercises, find the zeros and give the multiplicity of each. f(x) = 2x 4 (x 3 4x 2 + 4x)
-
Label each of the following statements true, false, or uncertain. Explain briefly. a. If the nominal exchange rate is fixed, the real exchange rate is fixed. b. When domestic inflation equals foreign...
-
Consider a country operating under fixed exchange rates. The IS curve is given by equation (20.1) a. Explain the term \(\left(i^{*}-\pi^{e}ight)\). Why does the foreign nominal interest rate appear...
-
Policy choices when the real exchange rate is "too high" and the nominal exchange rate is fixed An overvalued real exchange rate is a rate such that domestic goods are too expensive relative to...

Study smarter with the SolutionInn App


