The Hoechst process for ibuprofen synthesis, developed by the Boots-Hoechst-Celanese company, was a significant improvement over...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
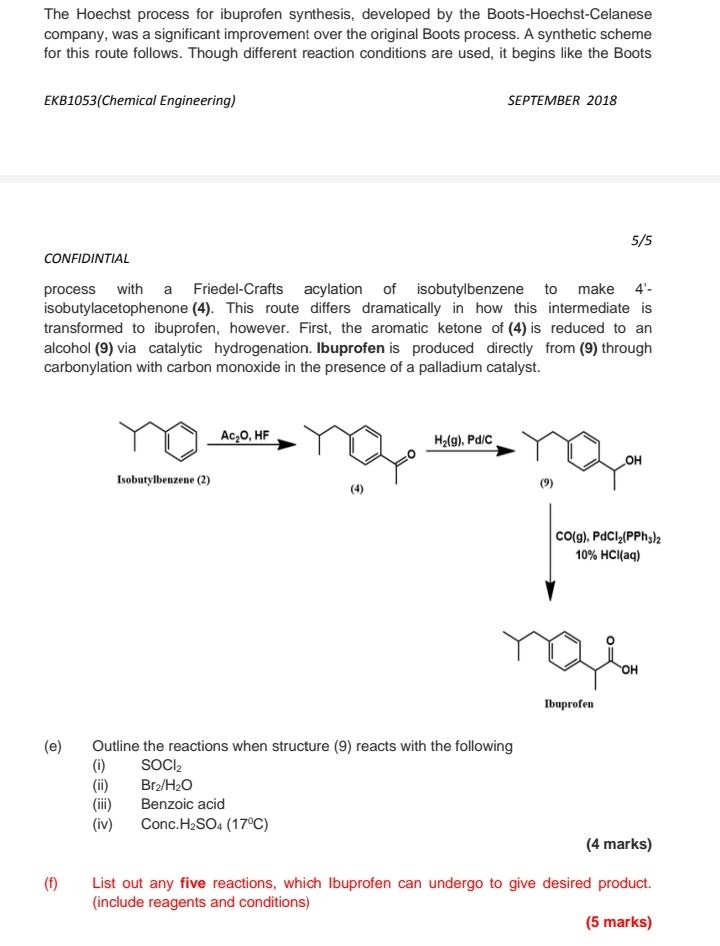
The Hoechst process for ibuprofen synthesis, developed by the Boots-Hoechst-Celanese company, was a significant improvement over the original Boots process. A synthetic scheme for this route follows. Though different reaction conditions are used, it begins like the Boots EKB1053(Chemical Engineering) SEPTEMBER 2018 5/5 CONFIDINTIAL process with a Friedel-Crafts acylation of isobutylbenzene to make 4'- isobutylacetophenone (4). This route differs dramatically in how this intermediate is transformed to ibuprofen, however. First, the aromatic ketone of (4) is reduced to an alcohol (9) via catalytic hydrogenation. Ibuprofen is produced directly from (9) through carbonylation with carbon monoxide in the presence of a palladium catalyst. Ac,0, HF H2(g), Pd/C OH Isobutylbenzene (2) (9) Co(g), PdCl,(PPh,)2 10% HCI(aq) он Ibuprofen (e) Outline the reactions when structure (9) reacts with the following (i) (ii) (iii) (iv) SOCI, Bra/H2O Benzoic acid Conc.H2SO4 (17°C) (4 marks) (f) List out any five reactions, which Ibuprofen can undergo to give desired product. (include reagents and conditions) (5 marks) The Hoechst process for ibuprofen synthesis, developed by the Boots-Hoechst-Celanese company, was a significant improvement over the original Boots process. A synthetic scheme for this route follows. Though different reaction conditions are used, it begins like the Boots EKB1053(Chemical Engineering) SEPTEMBER 2018 5/5 CONFIDINTIAL process with a Friedel-Crafts acylation of isobutylbenzene to make 4'- isobutylacetophenone (4). This route differs dramatically in how this intermediate is transformed to ibuprofen, however. First, the aromatic ketone of (4) is reduced to an alcohol (9) via catalytic hydrogenation. Ibuprofen is produced directly from (9) through carbonylation with carbon monoxide in the presence of a palladium catalyst. Ac,0, HF H2(g), Pd/C OH Isobutylbenzene (2) (9) Co(g), PdCl,(PPh,)2 10% HCI(aq) он Ibuprofen (e) Outline the reactions when structure (9) reacts with the following (i) (ii) (iii) (iv) SOCI, Bra/H2O Benzoic acid Conc.H2SO4 (17°C) (4 marks) (f) List out any five reactions, which Ibuprofen can undergo to give desired product. (include reagents and conditions) (5 marks)
Expert Answer:

Related Book For 

Introduction to Statistical Quality Control
ISBN: 978-1118146811
7th edition
Authors: Douglas C Montgomery
Posted Date:
Students also viewed these chemical engineering questions
-
A compound A (C6H) undergoes catalytic hydrogenation over Lindlar catalyst to give a compound B, which in turn undergoes ozonolysis followed by workup with aqueous H2O2 to yield succinic acid and two...
-
Like nickel, iron reacts with carbon monoxide to form a compound having the formula M (CO)n that obeys the 18-electron rule. What is the value of n in the formula Fe (CO)n?
-
Like nickel, iron reacts with carbon monoxide to form a compound having the formula M(CO)n that obeys the 18-electron rule. What is the value of n in the formula Fe(CO)n?
-
The control features of a bank account do not include: (a) having bank auditors verify the correctness of the bank balance per books. (b) minimizing the amount of cash that must be kept on hand. (c)...
-
The Heat Index HI, calculated from the air temperature and relative humidity, is the apparent temperature felt by the body. An equation used by the National Weather Service for calculating the HI is...
-
Telfer, Inc. reported net income of $2.1 million in 2017. Depreciation for the year was $155,800, accounts receivable decreased $358,900, and accounts payable decreased $283,400. Compute net cash...
-
What is the unknown isotope in the following fission reaction: \(\mathrm{n}+{ }^{235} \mathrm{U} ightarrow{ }^{131} \mathrm{I}+\) ? + \(3 \mathrm{n}\) A. \({ }^{86} \mathrm{Rb}\) B. \({ }^{102}...
-
Royal Jewels, a retail business, started business on June 25, 2019. It keeps a $300 change fund in its cash register. The cash receipts for the period from June 25 to June 30, 2019 are below....
-
All traffic between each branch office should be encrypted automatically. Which method of encryption is best for LAN - LAN encryption? What is necessary to set this up ?
-
1. 3. 5. 7. 9. 11. passive : change:: active: O movement O increase O flight stagnation convent: cloister :: eyrie: O hide nest O open show rotate: energy :: stop: O brake Ostart Orun push fumble:...
-
The general model for calculating a quantity variance is: Multiple Choice Actual price x (Actual quantity of inputs used Standard quantity allowed for output). Actual quantity of inputs used x...
-
Respond to the below questions using the following data for EVM: PV: 500,000 EV: 350,000 AC: 550,000 BAC: 1,200,000 Solve: 1) Compute SV. 2) Compute CV. 3) Compute SPI. 4) Compute CPI. 5) What does...
-
Understand how proliferated value-based payments are across the United States. Which is more prevalent today?
-
Present strategic recommendations for following business areas in Under Armour. 1) Direct to customer sales 2) Healthcare App (MapMyFitnesss)
-
Please create an IMC (integrated marketing communication) on Pratt&Whitney, mainly on in-person promotion/personal selling, events/meetings it attended(exhibition, military expo, exchange meetings...
-
Topics: Recursion and Merge-sort Problem Description Please make sure to read the document fully before starting! In this assignment, you will be tasked with writing methods that will solve some...
-
1. What is the difference between direct quote and indirect quote of foreign exchange? 2. What is a vehicle currency? What currency currently serves as the world's primary vehicle currency? 3. What...
-
You are planning to purchase your first home five years from today. The required down payment will be $50,000. You currently have $20,000. but you plan to contribute $500 each quarter to a special...
-
A random sample of 500 connecting rod pins contains 65 nonconforming units. Estimate the process fraction nonconforming. (a) Test the hypothesis that the true fraction defective in this process is...
-
Show that if the process is in control at the level , the exponentially weighted moving average is an unbiased estimator of the process mean.
-
There is a typographical error in the hypotheses (should be 22) in early printings of the 7th edition; this solution is for the correct hypotheses. Develop a test for the hypotheses H0 : 1 22 H1 : 1...
-
The adjusted trial balance columns in the worksheet of Elliot Painting Services are as follows. Required (a) Complete the worksheet. (b) Prepare the closing entries necessary at 30 June 2019,...
-
Michael Rau founded Michaels Fishing Supplies on 1 July 2019. The adjusted trial balance at 30 June 2020 (the end of the financial year) is shown below. Required (a) Prepare closing entries to be...
-
You are provided with the worksheet for Norman Pty Ltd for the year ended 30 June 2019. The adjustments have already been made and the worksheet begins with the adjusted trial balance. Assume all...

Study smarter with the SolutionInn App


