The irreversible first-order (wrt partial pressure of A) gas-phase reaction: A B is carried out...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
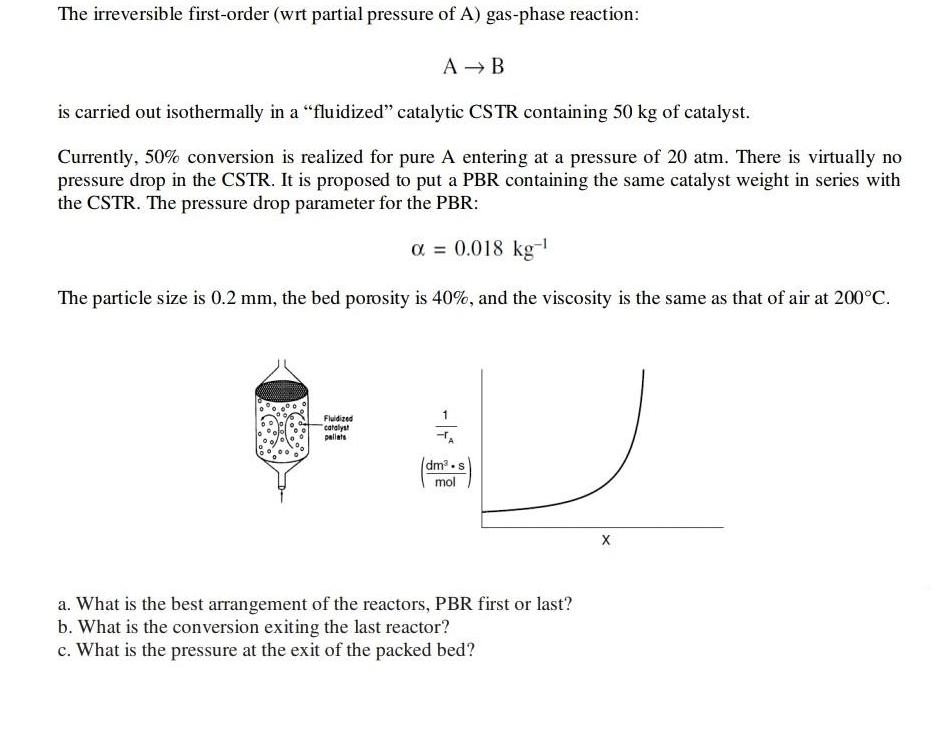
The irreversible first-order (wrt partial pressure of A) gas-phase reaction: A → B is carried out isothermally in a "fluidized" catalytic CSTR containing 50 kg of catalyst. Currently, 50% conversion is realized for pure A entering at a pressure of 20 atm. There is virtually no pressure drop in the CSTR. It is proposed to put a PBR containing the same catalyst weight in series with the CSTR. The pressure drop parameter for the PBR: α = 0.018 kg-¹ The particle size is 0.2 mm, the bed porosity is 40%, and the viscosity is the same as that of air at 200°C. Fluidized catalyst pallats -TA dm³-s mol X a. What is the best arrangement of the reactors, PBR first or last? b. What is the conversion exiting the last reactor? c. What is the pressure at the exit of the packed bed? The irreversible first-order (wrt partial pressure of A) gas-phase reaction: A → B is carried out isothermally in a "fluidized" catalytic CSTR containing 50 kg of catalyst. Currently, 50% conversion is realized for pure A entering at a pressure of 20 atm. There is virtually no pressure drop in the CSTR. It is proposed to put a PBR containing the same catalyst weight in series with the CSTR. The pressure drop parameter for the PBR: α = 0.018 kg-¹ The particle size is 0.2 mm, the bed porosity is 40%, and the viscosity is the same as that of air at 200°C. Fluidized catalyst pallats -TA dm³-s mol X a. What is the best arrangement of the reactors, PBR first or last? b. What is the conversion exiting the last reactor? c. What is the pressure at the exit of the packed bed?
Expert Answer:
Answer rating: 100% (QA)
Step 1 of 4 a The irreversible gasphase reaction is as follows A B The re... View the full answer

Related Book For 

Introduction to Chemical Engineering Thermodynamics
ISBN: 978-0071247085
7th edition
Authors: J. M. Smith, H. C. Van Ness, M. M. Abbott
Posted Date:
Students also viewed these physics questions
-
The irreversible first-order (wrt partial pressure of A) gas-phase reaction A B is carried out isothermally in a "fluidized" catalytic CSTR containing 50 kg of catalyst. Currently, 50% conversion is...
-
The production of maleic anhydride by the air oxidation of benzene was studied using a vanadium pentoxide catalyst [Chem Eng Sci, 43, 1051 (1988)]. The reactions that occur are C6H6 + 9/ 2 O2 C4H2O3...
-
1) A gas phase reaction A B takes place in a packed bed reactor. The pressure gradient arised along the length of PBR is assumed to be constant as dP / dW = -0.2 atm.kg. The conversion achieved in...
-
What are the lessons of the 1997 Asian financial crisis? Were the IMF supported adjustment programs successful in their objective? Were there flaws in their design?
-
First, select columns B through F and reformat them with the word wrap, so you can easily see the actual comments people sent in.
-
In Exercises find dy/dx by implicit differentiation. x = sec 1 y
-
Consider the regression model (8.8) described in Example 8.3 Graph the response function for this model and indicate the role the model parameters play in determining the shape of this function....
-
Amortization of Discount Stacy Company issued five-year, 10% bonds with a face value of $10,000 on January 1, 2010. Interest is paid annually on December 31. The market rate of interest on this date...
-
Reporting Issuance and Retirement of Long-Term Debt On the basis of the details of the following bonds payable and related discount accounts, indicate the items to be reported in the Financing...
-
The proposed rates were not in the range the CEO expected given the pricing analysis. The CEO has asked the pricing actuary to verify the total projected loss cost excluding potential large storm...
-
Was there a distinct American culture, society, or people by the end of 1750? Please cite specific examples from at least three different colonies, highlighting differences or similarities. How...
-
Argue that a limitation of damages provision should not be considered valid.
-
Draft an employment contract for yourself as a paralegal working for the firm of Pratt & Chase, a partnership of 25 attorneys working in your town. The contract is for a two-year period.
-
Consider the assets and liabilities associated with the experience of business failure. Should entrepreneurs who have experienced business failure try again?
-
This case arises out of a consent judgment entered by the district court in 1994 in which Gould, Inc. agreed to pay for environmental investigation and remediation of property sold by Gould, Inc. to...
-
Discuss the different challenges and benefits facing entrepreneurs who build portfolios of businesses concurrently in contrast to serial entrepreneurs who own one business at a time.
-
7. Determine whether the following codes are uniquely decodable: (a) (0,01, 11, 111) (b) (0,01, 110, 111} (c) (0, 10, 110, 111} (d) {1, 10, 110, 111}
-
Derive Eq. (18.33) from Eq. (18.32).
-
A stream of propane gas is partially liquefied by throttling from 200 bar and 370 K to 1 bar. What fraction of the gas is liquefied in this process? The vapor pressure of propane is given by Kq....
-
Assuming the validity of the De Priester charts, make the following VLE calculations far the ethane (1)/propane(2)/isobutane(3)/isopentane(4) system: (a) BVBL P, given x1 = 0.10, x2 = 0.20. x3 =...
-
Wet steam at 1,100 kPa expands at constant enthalpy (as in a throttling process) to 101.33 kPa, where its temperature is 105oC. What is the quality' of the steam in its initial state?
-
Reconsider the data of Problem 31. Data from Problem 31 The following three investment opportunities are available. The returns for each investment for each year vary, but the first cost of each is...
-
The following three investment opportunities are available. The returns for each investment for each year vary, but the first cost of each is $20,000. Based on a future worth analysis, which...
-
Explain the concept of a control variable and the assumption necessary for a control variable to be effective.

Study smarter with the SolutionInn App


