The mass percent of KHP in an unknown salt sample was determined using both volumetric and...
Fantastic news! We've Found the answer you've been seeking!
Question:

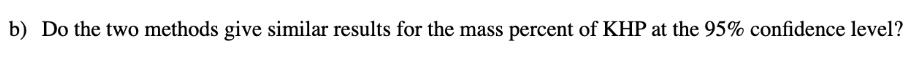
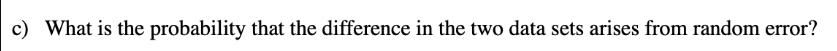
Transcribed Image Text:
The mass percent of KHP in an unknown salt sample was determined using both volumetric and gravimetric titration. Ten determinations using each method were performed: Volumetric (% KHP): 34.22 33.86 34.07 34.49 34.69 33.71 34.28 34.11 34.26 34.10 34.18 34.32 34.11 34.33 34.17 34.12 Gravimetric (% KHP): 34.35 34.26 34.10 34.19 a) Do the two methods give similar standard deviations? b) Do the two methods give similar results for the mass percent of KHP at the 95% confidence level? c) What is the probability that the difference in the two data sets arises from random error? The mass percent of KHP in an unknown salt sample was determined using both volumetric and gravimetric titration. Ten determinations using each method were performed: Volumetric (% KHP): 34.22 33.86 34.07 34.49 34.69 33.71 34.28 34.11 34.26 34.10 34.18 34.32 34.11 34.33 34.17 34.12 Gravimetric (% KHP): 34.35 34.26 34.10 34.19 a) Do the two methods give similar standard deviations? b) Do the two methods give similar results for the mass percent of KHP at the 95% confidence level? c) What is the probability that the difference in the two data sets arises from random error?
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
A couple has two children. What is the probability that both are boys, given that the first is a boy?
-
What is the probability that an electron will jump across the energy gap Eg in a diamond that has a mass equal to the mass of Earth? Use the result of Sample Problem 41-1 and the molar mass of carbon...
-
What is the probability that the selected person responded or is in the 18-21 age bracket? Age 18-21 22-29 255 20 30-39 40-49 136 16 50-5960 and over Responded 73 245 138 202 Refused 27 49
-
A room is 6 m by 5 m by 3 m. (a) If the air pressure in the room is 1 atm and the temperature is 300 K, find the number of moles of air in the room. (b) If the temperature rises by 5 K and the...
-
Prepare journal entries for the following selected transactions of Dulcinea Company for 2012. 2012 Dec. 13 Accepted a $ 9,500, 60-day, 8% note dated December 13 in granting Miranda Lee a time...
-
A delivery of 25 parts is subject to a quality control according to the following scheme: A sample of size 5 is drawn (without replacement of drawn parts). If at least one part is faulty, then the...
-
A long rotor can be balanced by adding weights in a. a single plane b. any two planes c. two specific planes
-
Bailey Corporation?s financial statements (dollars and shares are in millions) are provided here. a. What was net working capital for 2007 and 2008? b. What was Bailey?s 2008 free cash flow? c....
-
What are the most effective strategies for de-escalating high-tension conflicts, and what psychological principles underpin these techniques ?
-
You have been assigned the task of putting together a statement for the ACME Company that shows its expected inflows and outflows of cash over the months of July 2013 through December 2013. You have...
-
Solve the initial value problem: 2yy?+4=y2+4x2yy?+4=y2+4x withy(0)=5y(0)=5. 1 point) Solve the initial value problem \( 2 y y^{\prime}+4=y^{2}+4 x \) with \( y(0)=5 \). a. To solve this, we should...
-
Make a list of the changes you should make in the next 30 days to improve your Aji Amarillo Paste point-of-sale packaging.
-
Stationery Supplies uses a 24 percent annual interest rate to represent the cost of capital, and pays a liability insurance for 2% monthly rate of the value of the office supplies. The holding cost...
-
What is the reported on a TRICARE CMS-1500 claim in Block 1a?
-
The following is a design matrix X, response vector Y and (XX) for a multiple linear regression 3.1 Find X'Y. 1 0 1 1 02 1 0 3 X = Y = 1 1 1 1 2 52 to 1 3 (x'x) 1.33 -0.33 0.67 -0.50 0.00 -0.33 -0.50...
-
Given this question-Cumulative earnings or Y-T-D earning for Billy Bob is $5872.53 for the week ending April 17 of the current year. Billy Bob is married with five dependents. Social security is 6.2%...
-
As Patrick Sportwear Co. completes plans for its new assembly line of Liverpool, it identifies 30 different tasks in the production process. VP of Operations Kevin Keegan now faces the job of...
-
In Exercises find dy/dx by implicit differentiation. xy - y = x
-
Hydrogen sulfide can be removed from natural gas by the reaction 2H2S(g) + SO2(g) 3S(s) + 2H2O(g) Calculate Go and K (at 298 K) for this reaction. Would this reaction be favored at a high or low...
-
Calculate w and E when 1 mole of a liquid is vaporized at its boiling point (80oC) and 1.00 atm pressure. Hvap for the liquid is 30.7 kJ mol21 at 80.oC.
-
One of the few industrial scale processes that produces organic compounds electrochemically is used by the Monsanto Company to produce 1,4 dicyanobutane. The reduction reaction is 2CH2PCHCN + 2H+ +...
-
The W14 \(\times 26\) structural A-36 steel member is used as a 20 -ft-long column that is assumed to be fixed at its top and fixed at its bottom. If the 15-kip load is applied at an eccentric...
-
The W14 \(\times 26\) structural A-36 steel member is used as a column that is assumed to be fixed at its top and pinned at its bottom. If the 15-kip load is applied at an eccentric distance of 10...
-
Determine the maximum eccentric load \(P\) the 2014-T6aluminum-alloy strut can support without causing it either to buckle or yield. The ends of the strut are pin connected. a $150 mm 150 mm 100 mm...

Study smarter with the SolutionInn App


