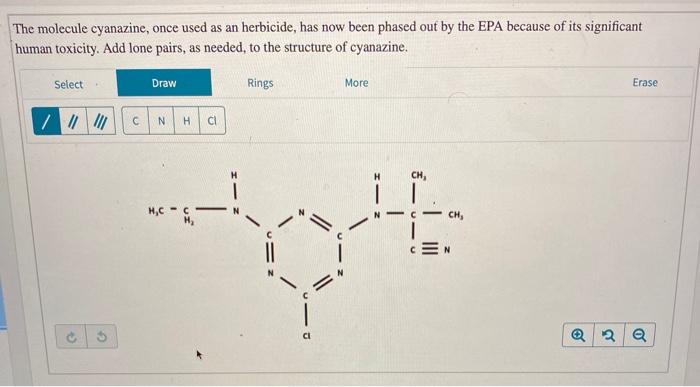
The molecule cyanazine, once used as an herbicide, has now been phased out by the EPA...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
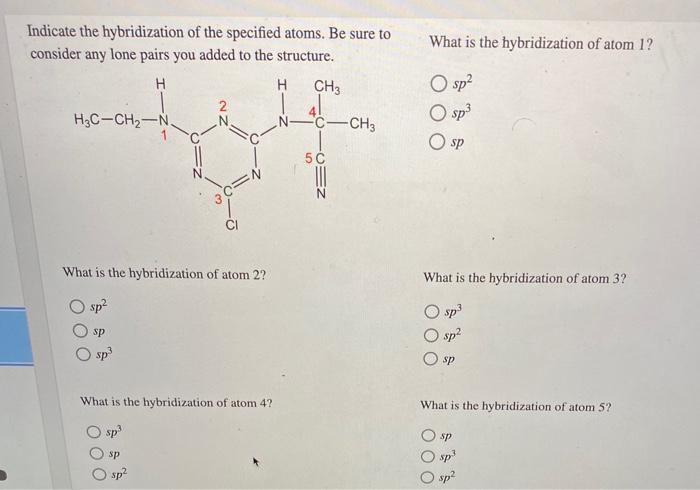
The molecule cyanazine, once used as an herbicide, has now been phased out by the EPA because of its significant human toxicity. Add lone pairs, as needed, to the structure of cyanazine. Select Draw Rings More Erase H H H,C C - CH, Indicate the hybridization of the specified atoms. Be sure to consider any lone pairs you added to the structure. What is the hybridization of atom 1? O sp? O sp3 H CH3 H3C-CH2-N N C-CH3 O sp What is the hybridization of atom 2? What is the hybridization of atom 3? sp2 O sp O sp? sp sp sp What is the hybridization of atom 4? What is the hybridization of atom 5? O sp O sp sp sp sp2 O sp O 00 O 00 The molecule cyanazine, once used as an herbicide, has now been phased out by the EPA because of its significant human toxicity. Add lone pairs, as needed, to the structure of cyanazine. Select Draw Rings More Erase H H H,C C - CH, Indicate the hybridization of the specified atoms. Be sure to consider any lone pairs you added to the structure. What is the hybridization of atom 1? O sp? O sp3 H CH3 H3C-CH2-N N C-CH3 O sp What is the hybridization of atom 2? What is the hybridization of atom 3? sp2 O sp O sp? sp sp sp What is the hybridization of atom 4? What is the hybridization of atom 5? O sp O sp sp sp sp2 O sp O 00 O 00
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Chloroform, CHCl3, a volatile liquid, was once used as an anesthetic but has been replaced by safer compounds. Chloroform boils at 61.7C and has a heat of vaporization of 31.4 kJ/mol. What is its...
-
Chloroform, CHCl 3 , a volatile liquid, was once used as an anesthetic but has been replaced by safer compounds. Chloroform boils at 61.7C and has a heat of vaporization of 31.4 kJ/mol. What is its...
-
Once it has been started by sufficient suction, the siphon in Fig. P3.169 will run continuously as long as reservoir fluid is available. Using Bernoullis equation with no losses, show (a) that the...
-
(a) Victor and Maria regularly buy and sell a number of items on eBay, Craig's List, and through the free community newspaper, from which they earn about $4,000 each year. What is the accumulated...
-
Discuss the role music plays in advertising. Why might companies such as Microsoft, Cadillac, and Nike pay large sums of money for the rights to use popular songs in their commercials?
-
Since its founding in 2010, the Chinese communications technology company Xiaomi has established itself as a true competitor among both global incumbents like Apple and Samsung and local niche...
-
A single cylinder, single acting pump lifts 30 litres of water per second against a static head of 25 m. The cylinder bore is 350 mm and stroke is 450 mm. Determine the speed of pump if slip is 2...
-
Below are eight common methods that project and functional employees can use to provide communications: a. Counseling sessions e. Project office memo b. Telephone conversation f. Project office...
-
Peter Smith was the owner and operator of Long Arm Guns based in Nashville, TN. Peter had bought and sold guns legally for years as a licensed arms merchant. In January of 2022, Peter sold an Ak47...
-
Jamie Lee Jackson, age 26, is in her last semester of college and is anxiously waiting for graduation day that is just around the corner! She still works part-time as a bakery clerk, has been...
-
Pinder Ltd is considering a merger with Value Co , which will exchange its 2 shares for 5 shares of Value Co . Pinder Ltd ' s share price is $ 3 4 . 3 2 and Value Co's share price is $ 1 1 . 9 8 ....
-
what is the impact of gender stereotypes and gender role assignment on gender development in young children? How has our concept of gender identity changed and broadened in recent years ?
-
By what percentage does the purchasing power of the currency decrease in five years, if it is reduced by 10% per year?
-
Listen to "Observations in Early Childhood Education" (The Everything ECE Podcast, 2023, 10.5 minutes): Journal Prompt(s): How did the podcast episode deepen your understanding of the role of...
-
Leveraged loans typically have A. Fixed Interest rates B. Variable interest rates Explain both rates briefly.
-
As a financial analyst specializing in the aerospace industry for USC Pension & Investment Inc., you are asked by your managing partner, Mr. Adam Smith, to evaluate the potential of this new GPWS...
-
Maple Co. provides for bad debts expense at the rate of 2.89% of credit sales for the period. On Jan 1, 20X1, the Allowance for Bad Debts was $9,000. There were $16,000 of accounts written off during...
-
What is EBIT/eps analysis? What information does it provide managers?
-
a. Consider a metal atom, which we will give the symbol M. Metal M can readily form the M+ cation. If the sphere on the left below represents the metal atom M, which of the other three spheres would...
-
A 50.0-mL volume of AgNO3 solution contains 0.0285 mol AgNO3 (silver nitrate). What is the molarity of the solution?
-
If the n quantum number of an atomic orbital is 4, what are the possible values of l? If the l quantum number is 3, what are the possible values of ml?
-
When and why should exchange rates change under a fixed-exchange-rate system?
-
Using the diagram in exercise 5, illustrate the effect of the change in Japanese tastes if exchange rates are fixed. What will happen to the foreign exchange market equilibrium? Data from in exercise...
-
How does a currency speculator profit from exchange-rate changes? Give an example of a profitable speculation.

Study smarter with the SolutionInn App


