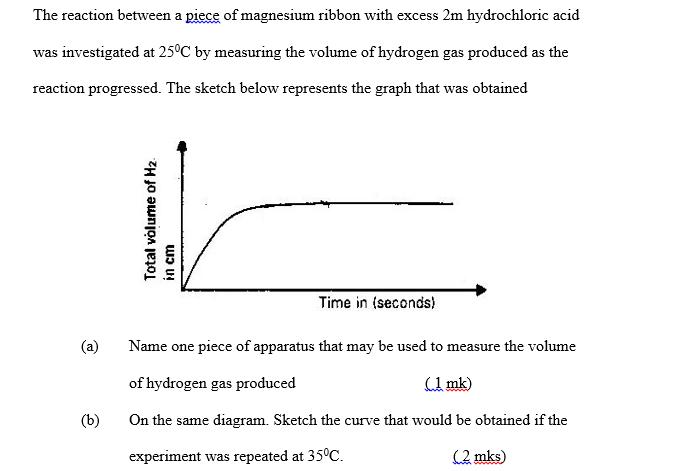
The reaction between a piece of magnesium ribbon with excess 2m hydrochloric acid was investigated at...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
The reaction between a piece of magnesium ribbon with excess 2m hydrochloric acid was investigated at 25°C by measuring the volume of hydrogen gas produced as the reaction progressed. The sketch below represents the graph that was obtained (a) (b) Total volume of H₂ M cm Time in (seconds) Name one piece of apparatus that may be used to measure the volume of hydrogen gas produced (1 mk) On the same diagram. Sketch the curve that would be obtained if the experiment was repeated at 35°C. (2 mks) The reaction between a piece of magnesium ribbon with excess 2m hydrochloric acid was investigated at 25°C by measuring the volume of hydrogen gas produced as the reaction progressed. The sketch below represents the graph that was obtained (a) (b) Total volume of H₂ M cm Time in (seconds) Name one piece of apparatus that may be used to measure the volume of hydrogen gas produced (1 mk) On the same diagram. Sketch the curve that would be obtained if the experiment was repeated at 35°C. (2 mks)
Expert Answer:
Answer rating: 100% (QA)
a A gas syringe is one tool that can be used to gauge the amount of hydrogen created during the reac... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
The Hall effect can be used to measure blood flow rate because the blood contains ions that constitute an electric current. (a) Does the sign of the ions influence the emf? Explain. (b) Determine the...
-
The basic barometer can be used to measure the height of a building. If the barometric readings at the top and at the bottom of a building are 730 and 755 mm Hg, respectively, determine the height of...
-
The Michelson interferometer can be used to measure the index of refraction of a gas by placing an evacuated transparent tube in the light path along one arm of the device. Fringe shifts occur as the...
-
Instructions In simplified form, you are going to be producing a script. The script will create 2 tables, load the 2 tables with data and then using PL/SQL it will process those 2 tables and with 2...
-
1. The court discusses the essential facilities doctrine. What do you suppose the plaintiffs were alleging the essential facility at issue here was? Why did their argument fail? 2. Do you agree that...
-
Haskins and Jones, Attorneys-at-Law, maintains its books on a cash basis. During 2021, the law firm collected $545,000 for services rendered to its clients and paid out $412,000 in expenses. You are...
-
Explain parameters and return values in the framework of messages. Provide examples.
-
The lost Israeli soldier mentioned in Problem 2-21 decides to rest for a few minutes before entering the desert oasis he has just found. Closing his eyes, he dozes off for 15 minutes, wakes, and...
-
Find the average value fave of the function f on the given interval. f(x) = x (x3+10) 2' [-1, 1] fave = | X
-
Barlow Company manufactures three products: A, B, and C. The selling price, variable costs, and contribution margin for one unit of each product follow: The same raw material is used in all three...
-
Explain in six-page detail why the recent talk of not teaching African American History is school an unethical act. Explain and provide a background of the facts, an analysis of the dilemma,...
-
What is the best reason for marking an existing static method private within in an interface? A. It allows the method to be overridden in a subclass. B. It hides the secret implementation details...
-
What is the output of the following application? A. Spoiled! B. Yummy! C. The application completes without printing anything. D. One line of this application fails to compile. E. Two lines of this...
-
Which statement about the following interface is correct? A. The code compiles. B. The method enterAtmosphere() does not compile. C. The method getCircumference() does not compile. D. The method...
-
How many of the classes in the figure can write code that references the sky() method? A. None B. One C. Two D. Three E. Four. com.color Red protected boolean light Blue void sky () com.light Bright...
-
Given the following class declaration, which options correctly declare a local variable containing an instance of the class? A. new Sunset() extends Sky {}; B. new Sky(); C. new Sky() {} D. new Sky()...
-
On September 1, 2021, PQR Corporation purchased a new machine on a deferred payment basis. A down payment of P20,000 was made and 4 annual installments of P60,000 each are to be made beginning on...
-
Per Bag Direct materials: 25 pounds of CWhiz-2000 @ $0.08/lb. = $ 2.00 Direct labor: 0.05 hour @ $32.00/hr. = $ 1.60 The company manufactured 100,000 bags of Cheese-Be-Good in December and used...
-
Answer the following questions without referring to Table 2.1: (a) What are the main subatomic particles that make up the atom? (b) What is the relative charge (in multiples of the electronic charge)...
-
Acetone, CH3COCH3, is a nonelectrolyte; hypochlorous acid, HClO, is a weak electrolyte; and ammonium chloride, NH4Cl, is a strong electrolyte.
-
What is the pH at 25 C of water saturated with CO 2 at a partial pressure of 1.10 atm? The Henry's law constant for CO 2 at 25 C is 3.1 10 2 mol/L-atm. The CO 2 is an acidic oxide, reading with H 2...
-
In Fig. 1130, there is no explicit invariant that states that a Transaction cannot be linked to movements that are linked to the same account, because the structure of the model already demands is....
-
Apply the Account/Transaction pattern to the parallel financial transactions of the example shown in Figs. 1131 to 1135. As the bills may be paid forward, it may be necessary to use memo entries....
-
Look at Fig. 1119 and create a new discount strategy that gives a percentage of discount for sales with more than a certain number of products. Order | < > < > +nr: Natural = < > +date: Date...

Study smarter with the SolutionInn App


