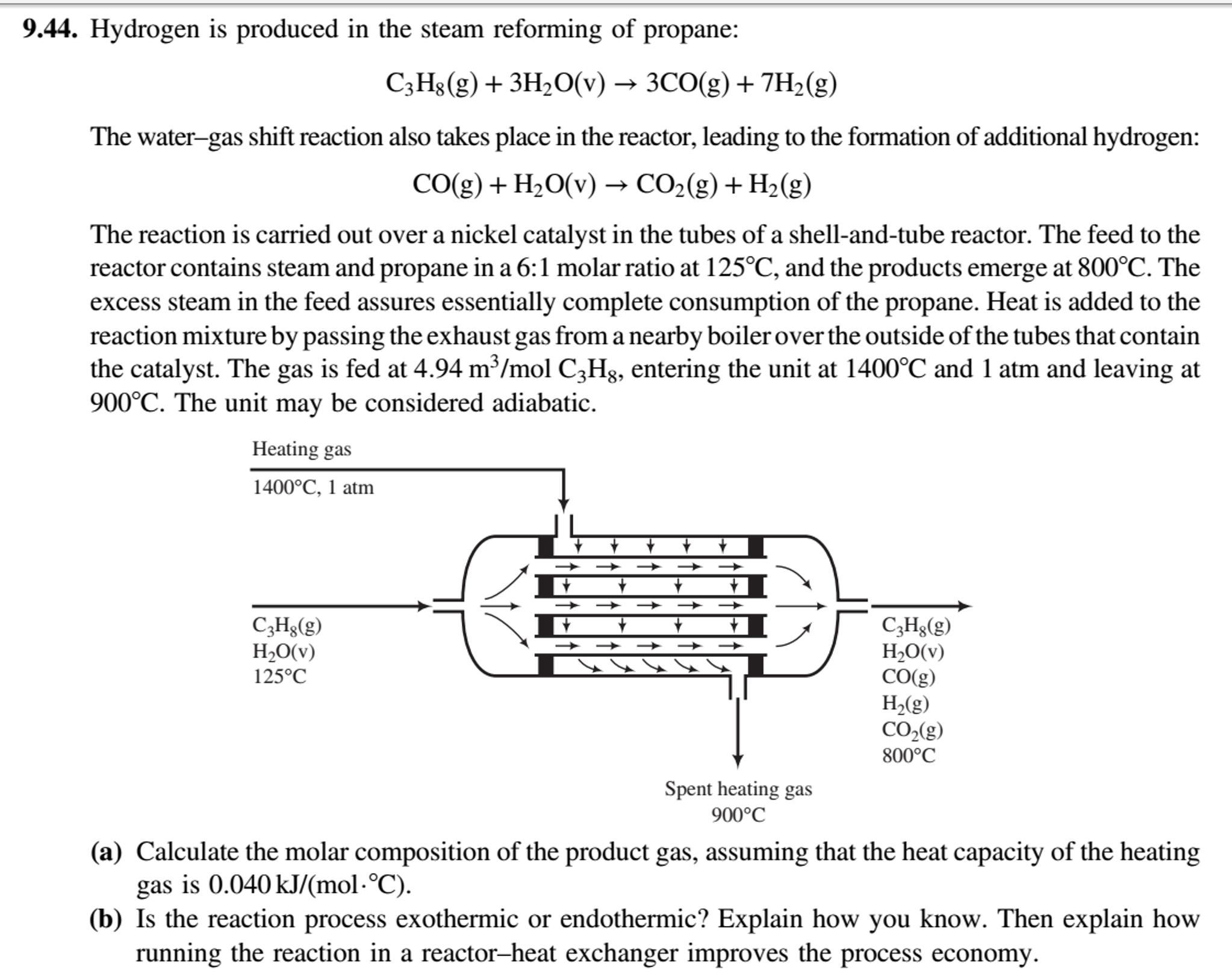
9.44. Hydrogen is produced in the steam reforming of propane: C3H8(g) + 3H2O(v) 3CO(g) +...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
9.44. Hydrogen is produced in the steam reforming of propane: C3H8(g) + 3H2O(v) → 3CO(g) + 7H2(g) The water-gas shift reaction also takes place in the reactor, leading to the formation of additional hydrogen: CO(g) + H2O(v) → CO2(g) + H2(g) The reaction is carried out over a nickel catalyst in the tubes of a shell-and-tube reactor. The feed to the reactor contains steam and propane in a 6:1 molar ratio at 125°C, and the products emerge at 800°C. The excess steam in the feed assures essentially complete consumption of the propane. Heat is added to the reaction mixture by passing the exhaust gas from a nearby boiler over the outside of the tubes that contain the catalyst. The gas is fed at 4.94 m³/mol C3H3, entering the unit at 1400°C and 1 atm and leaving at 900°C. The unit may be considered adiabatic. Heating gas 1400°C, 1 atm C3Hg(g) H2O(v) 125°C C,Hg(g) H,O(v) CO(g) H2(g) CO,(g) 800°C Spent heating gas 900°C (a) Calculate the molar composition of the product gas, assuming that the heat capacity of the heating gas is 0.040 kJ/(mol·°C). (b) Is the reaction process exothermic or endothermic? Explain how you know. Then explain how running the reaction in a reactor–heat exchanger improves the process economy. 9.44. Hydrogen is produced in the steam reforming of propane: C3H8(g) + 3H2O(v) → 3CO(g) + 7H2(g) The water-gas shift reaction also takes place in the reactor, leading to the formation of additional hydrogen: CO(g) + H2O(v) → CO2(g) + H2(g) The reaction is carried out over a nickel catalyst in the tubes of a shell-and-tube reactor. The feed to the reactor contains steam and propane in a 6:1 molar ratio at 125°C, and the products emerge at 800°C. The excess steam in the feed assures essentially complete consumption of the propane. Heat is added to the reaction mixture by passing the exhaust gas from a nearby boiler over the outside of the tubes that contain the catalyst. The gas is fed at 4.94 m³/mol C3H3, entering the unit at 1400°C and 1 atm and leaving at 900°C. The unit may be considered adiabatic. Heating gas 1400°C, 1 atm C3Hg(g) H2O(v) 125°C C,Hg(g) H,O(v) CO(g) H2(g) CO,(g) 800°C Spent heating gas 900°C (a) Calculate the molar composition of the product gas, assuming that the heat capacity of the heating gas is 0.040 kJ/(mol·°C). (b) Is the reaction process exothermic or endothermic? Explain how you know. Then explain how running the reaction in a reactor–heat exchanger improves the process economy.
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The following reaction is carried out at 500 K in a container equipped with a movable piston. After the reaction has reached equilibrium, the container has the composition depicted here. Suppose the...
-
If the reaction is carried out at constant V, how does the total pressure change if T increases? H 2 (g) + Cl 2 (g) 2HCl(g) at equilibrium. Assume ideal gas behavior.
-
The following elementary homogeneous gas-phase reaction is carried out isothermally in an ideal, steady state 0.2 L plug flow reactor: 2 F --> P. PThe inlet molar feed consists of a 50%-50% mixture...
-
On May 1, Johnson Corporation purchased inventory for $40,000 on credit. On May 15, Johnson sold inventory with a cost of $10,000 for $25,000 on credit. Prepare journal entries to record these...
-
Euler also found the sum of the p-series with p = 4: Use Euler's result to find the sum of the series. (a) (b) (4) = 2,7 = 90 3
-
A long-term investment in bonds with a cost of $75,000 was sold for $84,000 cash. (a) What was the gain or loss on the sale? (b) What was the effect of the transaction on cash flows? (c) How should...
-
Define the following: a. Asset b. Liability c. Net asset
-
Refer to Problem 21. Assume that management desires a 12-day project completion time. a. Formulate a linear programming model that can be used to assist with the crashing decisions. b. What...
-
The interest rate is fixed at 10%. You are offered an asset that pays $1,000 from now until eternity at yearly intervals. You find its present value by calculating the sum of the discounted annual...
-
A spring with an unstrained length of 0.074 m and a spring constant of 2.4 N/m hangs vertically downward from the ceiling. A uniform electric field directed vertically upward fills the region...
-
6. Give an account of the relativistic theory of Doppler effect. 7. Compare and contrast Doppler effect observed in light and sound. 8. What is Doppler effect? Show that for a source receding along...
-
DELISH DESIGNS & EQUIPMENT COMPANY(DDEC) Delish Designs & Equipment Company (DDEC) is a medium-sized enterprise in the private sector. DDEC manufactures and sells a range of protective wear and...
-
1)Identifying and describing (3-4) psychological biases that may influence a person's financial decision-making, including insights as to why you may be susceptible to these biases? 2) Outline...
-
Explain why an ABC system may result in different cost allocations compared with a simple allocation basis such as direct labor hours. Identify any ethical issues or considerations that should be...
-
What elements of due diligence do you think would be most important to investigate and consider as part of the potential acquisition process?
-
Identify 2-3 systematic biases in group decision making. (a) Define each bias and explain how it can impair group decisions, and (b) outline some possible mitigation strategies to overcome each bias...
-
In 2021, the additional child tax credit for a married filing jointly taxpayer with 2 children: a. Is only available for children under the age of 6 b. Is $1,000 for children under the age of 6 c. Is...
-
What types of inventory issues Starbucks might reflect upon at the end of each year? The mission of Starbucks is to inspire and nurture the human spiritone person, one cup, and one neighborhood at a...
-
List two applications of freezing-point depression.
-
Why is it generally impossible to predict the rate law for a reaction on the basis of the chemical equation only?
-
Photographers hypo, used to fix negatives during the development process, is sodium thiosulfate pentahydrate. What is the chemical formula of this compound?
-
Approximately 50,000 new titles, including new editions, are published each year in the United States, giving rise to a $25 billion industry in 2001. In terms of percentage of sales, this industry...
-
When you think of political persuasion, you may think of the effortsthat political campaigns undertake to persuade you that their candidate is betterthan the other candidate. In truth, campaigns are...
-
Exeter, Inc., is a catalog firm that sells products in a number of different catalogs that it owns. The catalogs number in the dozens but fall into nine basic categories: 1. Clothing 2. Housewares 3....

Study smarter with the SolutionInn App


