The standard potentials of the two half-cells A++, A** | Pt and B**, B* | Pt...
Fantastic news! We've Found the answer you've been seeking!
Question:
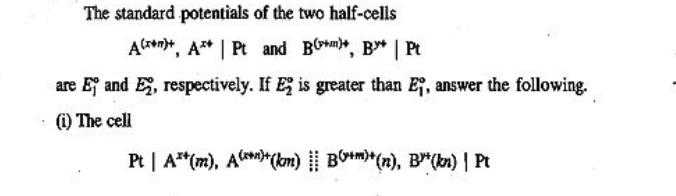

Transcribed Image Text:
The standard potentials of the two half-cells A++, A** | Pt and B**, B* | Pt are Ej and E, respectively. If E2 is greater than E, answer the following. (i) The cell Pt | A**(m), A()+(km) B+)+(n), B³(kn) | Pt was constructed, where k has some constant value. The cell was operated reversibly up to its exhaustion point, i.e. to a point where no more reaction in the cell takes place and hence no more current can be withdrawn from the cell. Show that (a) the half-cell potentials at the exhaustion point are given by the expression nE + ME (n + m) (b) The value of k at the exhaustion point at 298 K is given by the relation nm(E-E) (0.059 13 V)(m + n) logk (ii) Which of the following two cells will give a spontaneous cell reaction when k The standard potentials of the two half-cells A++, A** | Pt and B**, B* | Pt are Ej and E, respectively. If E2 is greater than E, answer the following. (i) The cell Pt | A**(m), A()+(km) B+)+(n), B³(kn) | Pt was constructed, where k has some constant value. The cell was operated reversibly up to its exhaustion point, i.e. to a point where no more reaction in the cell takes place and hence no more current can be withdrawn from the cell. Show that (a) the half-cell potentials at the exhaustion point are given by the expression nE + ME (n + m) (b) The value of k at the exhaustion point at 298 K is given by the relation nm(E-E) (0.059 13 V)(m + n) logk (ii) Which of the following two cells will give a spontaneous cell reaction when k
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The standard potentials of proteins are not commonly measured by the methods described in this chapter because proteins often lose their native structure and function when they react on the surfaces...
-
The standard potentials of lead and tin are 126 mV and 136 mV respectively at 25C, and the overvoltage for their deposition are close to zero. What should their relative activities be in order to...
-
At 298 K a cell reaction has a standard cell potential of +0.17 V. The equilibrium constant for the reaction is 5.5 105. What is the value of n for the reaction?
-
Discussion Post: Suppose you were considering investing in a Series I Bond. The bond will pay 5% interest. However, bond A compounds annually, and bond B compounds semiannually. Provide a detailed...
-
Your company has decided to pay employees for doing charity work. Employees can spend 1 hour working with a charitable on nonprofit group for every 40 they work. Employees will be paid for this hour,...
-
Suppose that the paper clip industry is perfectly competitive. Also assume that the market price for paper clips is 2 cents per paper clip. The demand curve faced by each firm in the industry is: a....
-
Figure 7.26 shows a cart striking a spring. In Figure 7.26a, consider the motion of the cart along the direct path from the initial position \(x_{1}\), which is the position at which the cart makes...
-
Given the following production schedule, compute the available-to-promisequantities. WEEK 4 Model B MPS 20 20 20 BI 20 20 20 20 Committed Customer Orders10 1010 1010 ATP:D 10
-
A call option on a single share of St. Margaret Beer Co.'s common stock has a market price of $7.02 and expires in six months. The option has an exercise, or strike, price of $68.00, and the current...
-
Presented here is the income statement for Big Sky Incorporated for the month of February Sales Cost of goods sold Gross profit Operating expenses Operating loss $ 61,500 51,300 $ 10,200 15,000 5...
-
1. Construct the circuit of Figure 3.1 using the following resistors: R-510 2, R1=1k, R-6.8k, R3-3.3k, R-5.1k, R,-2.k, Re-10k. R R2 R4 III 6 V R3 R5 Figure 3.1 Nodal analysis circuit. 2. Activate the...
-
Implement the following pseudo-code in MIPS. You need to define the vectors (8x and sy) and other variables (N) in the data section (under .data). All vectors are of N, which are initialized using a...
-
What are the numbers, percentages, and data specifics when it comes to domestic violence programs and domestic violence hotlines prior to COVID 19 and pre-pandemic in Hawaii? Please explain in huge...
-
a. Prepare a flexible budget for the actual units produced for May, June, and July in the Machining Department. Assume depreciation is a fixed cost. If required, use per unit amounts carried out to...
-
Suppose that the income from visits by foreign tourists increases (the rest remains constant), that is, they are higher than those indicated in the table for all years. For all years, briefly,...
-
system specifications: = 5 GHz fe B = 20MHz Consider a wireless LAN system with the follwoing HTX = 2 dB GRX = 2 dB Fading margin = 16 dB Path loss 90 dB PTX = 20 dBm TX losses: 3 dB Required SNR: 5...
-
for the given below vector use the expansion method to find their cross product. A=2i-3j+4k and B=1+2j+5k
-
Which of the companies has the lowest accounts receivable turnover in the year 20X2? a. Company A. b. Company B. c. Company C. d. CompanyD. 20X1 20X2 Credit Sales Average Receivables Balance $1.0...
-
Consult literature sources and list the observed timescales during which the following processes occur radiative decay of excited electronic states, molecular rotational motion, molecular vibrational...
-
The movement of atoms and ions on a surface depends on their ability to leave one position and stick to another, and therefore on the energy changes that occur. As an illustration, consider a...
-
Identify the systems for which it is essential to include a factor of 1/N on going from Q to q: (a) A sample of carbon dioxide gas, (b) A sample of graphite, (c) A sample of diamond, (d) Ice.
-
The primary problem with the hypothesis that life on Earth originated on Mars is that (a) Mars has never had water. (b) the proposed Martian fossils are much smaller than the tiniest bacteria on...
-
Which of these statements regarding the origin of life is false? (a) Life originated on an Earth whose atmosphere contained high levels of oxygen. (b)Miller and Urey obtained amino acids and other...
-
When you carry shopping bags, rather than grasp the handles with your hand as in Figure Q8.14a, you might choose to put them over your arm and slide the handle toward your elbow as in Figure Q8.14b....

Study smarter with the SolutionInn App



