The temperature, at which the number of molecules in the v= 1 is 1/e times that...
Fantastic news! We've Found the answer you've been seeking!
Question:
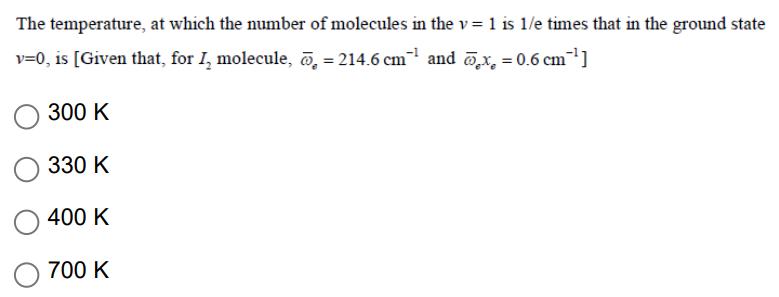
Transcribed Image Text:
The temperature, at which the number of molecules in the v= 1 is 1/e times that in the ground state v=0, is [Given that, for I molecule, 214.6 cm and @x=0.6 cm ] 300 K 330 K O 400 K 700 K The temperature, at which the number of molecules in the v= 1 is 1/e times that in the ground state v=0, is [Given that, for I molecule, 214.6 cm and @x=0.6 cm ] 300 K 330 K O 400 K 700 K
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these physics questions
-
In the case of gaseous nitrogen find: (a) The temperature at which the velocities of the molecules v1 = 300 m/s and v2 = 600 m/s are associated with equal values of the Maxwell distribution function...
-
The following bar graph represents the boiling temperature, the temperature at which a substance changes from liquid to gas at standard atmospheric pressure. Use this graph to answer below Exercises....
-
Consider a gas of diatomic molecules (moment of inertia I) at an absolute temperature T. If Eg is a ground-state energy and E ex is the energy of an excited state, then the Maxwell-Boltzmann...
-
What do you think Dan Barry means when he claims that Ring Dings taste like America?
-
A meter stick is held vertically with one end on the floor and is then allowed to fall. Find the speed of the other end just before it hits the floor, assuming that the end on the floor does not slip.
-
The principal stresses acting at a point on a thin-walled cylindrical pressure vessel are \(\sigma_{1}=p r / t, \sigma_{2}=p r / 2 t\), and \(\sigma_{3}=0\). If the yield stress is \(\sigma_{Y}\),...
-
Is it possible to provide absolute security for an organizations information sys tem? Why or why not?
-
Wallowa Company is considering a long-term investment project called ZIP. ZIP will require an investment of $120,000. It will have a useful life of 4 years and no salvage value . Annual revenues...
-
Garden Pro Corporation has sales of $4,437,338; income tax of $468,826; the selling, general and administrative expenses of $205,212; depreciation of $383,065; cost of goods sold of $2,623,466; and...
-
Company A is a global company based in the United States that operates in the financial industry. Company A serves its customers with financial products, such as checking accounts, bank cards, and...
-
Sampson Company uses a job order cost system with overhead applied to products based on direct labor hours. Based on previous history, the company estimated its total overhead for the coming year...
-
1. Which of the following is an example of a measurable outcome for the patient who has undergone a surgical procedure with a pain rating of 7 on a scale of 0 to 10? a.The patients pain will be under...
-
Explain the causes of conflict situation due to the style or nature of the work.
-
1.)The table below summarizes the expected impacts of a potential investment project. Complete the table by entering the taxable income and taxes for years 1 through 5, and NATCF values for years 0...
-
6. At an amusement park, first a child and then an adult are dropped down a slide (slide). Friction can be neglected. Which of them takes less time to reach the lower end? You can put any type of...
-
How can HR benefit from an effective usage of arbitration and collective bargaining in the workplace?
-
Factor the polynomial by grouping (if possible). 5n2p-10np 5n2 +10n
-
Time Solutions, Inc. is an employment services firm that places both temporary and permanent workers with a variety of clients. Temporary placements account for 70% of Time Solutions' revenue;...
-
How much energy does it take to convert 0.500 kg of ice at 220oC to steam at 250oC? Specific heat capacities: ice, 2.1 J g21 8C21; liquid, 4.2 Jg-1oC-1; steam, 2.0 Jg-1oC-1; Hvap = 40.7 kJ/ mol; Hfus...
-
Consider the following reaction at 248oC and 1.00 atm: CH3Cl(g) + H2(g) CH4(g) + HCl(g) For this reaction, the enthalpy change at 248oC is 283.3 kJ/ mol. At constant pressure the molar heat...
-
Suppose the reaction system UO2(s) + 4HF(g) UF4(g) + 2H2O(g) has already reached equilibrium. Predict the effect that each of the following changes will have on the equilibrium position. Tell...
-
Assuming that money is worth 10%, compute the present value of a. \($7,000\) received 15 years from today. Bing b. The right to inherit \($1,000,000\) 14 years from now. c. The right to receive...
-
For each of the following independent situations, compute the net after-tax cash flow amount by subtracting cash outlays for operating expenses and income taxes from cash revenue. The cash outlay for...
-
Gardner, Inc., plans to finance its expansion by raising the needed investment capital from the following sources in the indicated proportions and respective capital cost rates: Calculate the...

Study smarter with the SolutionInn App



