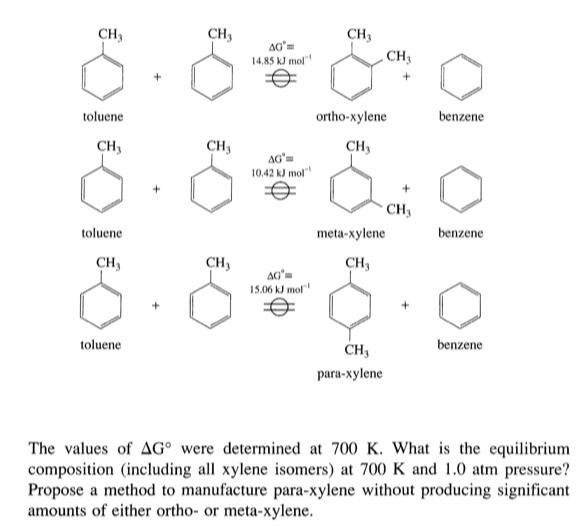
Xylene can be produced from toluene as written schematically: CH, CH3 CH3 AG 14,85 kJ mol CH3
Fantastic news! We've Found the answer you've been seeking!
Question:
Xylene can be produced from toluene as written schematically:

Transcribed Image Text:
CH, CH3 CH3 AG 14,85 kJ mol CH3 toluene ortho-xylene benzene CH, CH3 CH, 10.42 kJ mol" CH3 toluene meta-xylene benzene CH3 CH, CH, 15.06 kJ mol toluene CH, benzene para-xylene The values of AG° were determined at 700 K. What is the equilibrium composition (including all xylene isomers) at 700 K and 1.0 atm pressure? Propose a method to manufacture para-xylene without producing significant amounts of either ortho- or meta-xylene. CH, CH3 CH3 AG 14,85 kJ mol CH3 toluene ortho-xylene benzene CH, CH3 CH, 10.42 kJ mol" CH3 toluene meta-xylene benzene CH3 CH, CH, 15.06 kJ mol toluene CH, benzene para-xylene The values of AG° were determined at 700 K. What is the equilibrium composition (including all xylene isomers) at 700 K and 1.0 atm pressure? Propose a method to manufacture para-xylene without producing significant amounts of either ortho- or meta-xylene.
Expert Answer:
Answer rating: 100% (QA)
The equilibrium constant for the given reaction assuming ideal gas behavior can be calculated using ... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
What is the equilibrium composition of a reaction mixture if you start with 0.500 mol each of H2 and I2 in a 1.0-L vessel? The reaction is H2(g) + 12(g)--2HI(g) Kc = 49.7 at 458C
-
At 700 K the equilibrium constant for the reaction Is Kp= 0.76. A flask is charged with 2.00 atm of CCl4, which then reaches equilibrium at 700 K. (a) What fraction of the CCl4 is converted into C...
-
A 7.62 gram sample of xylene is burned to produce 25.3 g CO2 and 6.47 g H2O. If the molar mass is between 100 and 112 g/mol, what are the empirical and molecular formulas of xylene?
-
After 1 0 0 0 B . C . E . trade in Greece allowed for the accumulation of wealth by Kings Aristoi Farmers Foreigners
-
When and if Galileo dropped two balls from the top of the Leaning Tower of Pisa, air resistance was not really negligible. Assuming that both balls were of the same size, one made of wood and one of...
-
a. What is meant by a businesss underlying cost structure? b. Why is this information valuable to managers?
-
Based on the design, briefly discuss the data collection procedures to be used. Be sure to include the area rea of focus and targeted sample as part of these procedures. Develop a hypothetical...
-
The following events apply to Equipment Services Inc. in its first year of operation: 1. Acquired $60,000 cash from the issue of common stock. 2. Received a $8,200 cash advance for services to be...
-
The adjusted trial balance for Chiara Company as of December 31 follows. Cash Accounts receivable Interest receivable Debit $ 30,000 Credit 52,000 18,000 nt ok Notes receivable (due in 90 days)...
-
In December, Northern Kiteboarding magazine collected $80,000 for subscriptions for the next calendar year. The company collects cash in advance and then downloads the magazines to subscribers each...
-
If you invested $100,000 today and earned 7% per year, you would earn $100,000*0.07 = $7,000 in year 1. Say you invested for 30 years. How much would you earn in the last year, i.e year 30?
-
A case study analysis requires you to investigate a business problem, examine solutions and propose a recommendation based on research.. Prepare a case study for BP Texas City.
-
As an HR manager, you have heard rumors about potential efforts to unionize your warehouse employees. Using online resources, prepare a short list of guidelines (at least three points) hto help...
-
Consider the following information: State Probability ABC Inc.'s Return Boom 0.25 15% Normal 0.50 8% Slowdown 0.15 4% Recession 0.10 -3% i. 11. iii. What is the expected return? What is the variance?...
-
A case study analysis requires you to investigate a business problem, examine solutions and propose a recommendation based on research. Prepare a case study for Angelo's Pizza.
-
One of the 5 major tax issues associated with mergers, acquisitions and divestitures is whether the acquirer uses cash from newly issued equity or cash from retained earnings to purchase the target...
-
A toy rocket is launched upwards from the ground. It's initial upward acceleration is 5.0 m/s. The rocket only has enough fuel to supply a thrust for 10 seconds before it shuts off. (a) What is the...
-
A test car is driven a fixed distance of n miles along a straight highway. (Here n Z+.) The car travels at one mile per hour for the first mile, two miles per hour for the second mile, four miles...
-
A particular transition of the rubidium atom emits light whose frequency is 3.84 1014 Hz. (Hz is the abbreviation for hertz, which is equivalent to the unit /s, or s1.) Is this light in the visible...
-
Identify the phase transition occurring in each of the following. a. The water level in an aquarium tank falls continuously (the tank has no leak). b. A mixture of scrambled eggs placed in a cold...
-
What is the pH of a 0.35 M solution of methylammonium chloride, CH3NH3Cl? What is the concentration of methylamine in the solution?
-
Which transaction would generate foreign currency transaction exposure for Ambleu? A . Transaction 1 B . Transaction 2 C . Transaction 3
-
Based on Exhibit 1, what is the foreign exchange gain resulting from Transaction 3 on the December 31, 2016 fi nancial statements? A . NVK1.70 per ton B . NVK90.75 per ton C . NVK248.54 per ton
-
In response to the boards second question, Templeton should reply that if the change is made, the consolidated fi nancial statements for Consolidated Motors would begin to recognize: A . realized...

Study smarter with the SolutionInn App


