The vibrational food, and the anharmonicity constant up HD molecule are 3217cmt and 98 cmt respectively....
Fantastic news! We've Found the answer you've been seeking!
Question:
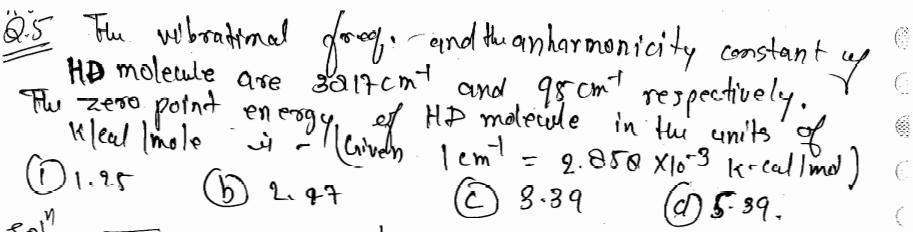
Transcribed Image Text:
The vibrational food, and the anharmonicity constant up HD molecule are 3217cmt and 98 cmt respectively. The Ter point energy of HD molecule in the units of zero Kleal = 1.25 (b) 2.27 (c) 5-39. (C) 8.39 The vibrational food, and the anharmonicity constant up HD molecule are 3217cmt and 98 cmt respectively. The Ter point energy of HD molecule in the units of zero Kleal = 1.25 (b) 2.27 (c) 5-39. (C) 8.39
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these physics questions
-
Using Table E.6, determine the lower-tail critical value for the Wilcoxon rank sum test statistic, T 1 , in each of the following one-tail tests: a. a = 0.05, n = 6, n = 8 b. a = 0.025, n = 6, n %3D...
-
The zero point energy of the particle in the box goes to zero as the length of the box approaches infinity. What is the appropriate analogue for the quantum harmonic oscillator?
-
The vibrational wave number of the oxygen molecule in its electronic ground state is 1580 cm3, whereas that in the first excited state (B 3L), to which there is an allowed electronic transition, is...
-
The pilot of a small boat charts a course such that the boat will always be equidistant from an upcoming rock and the shoreline. Describe the path of the boat. If the rock is 2 miles offshore, write...
-
The following information is available for Sentry Company for the first month of the year Revenues ....................................................$17,000 Expenses...
-
How many points did Ed score during the fourth game? A. 12 B. 11 C. 10 D. 9 E. 8 Jay's points 20 16 25 15 14 Ed's points 15 21 17
-
Selected accounts and a section of a worksheet for Sunhill Stores are shown below. Required (a) The beginning and ending inventory were \($45\) 760 and \($52\) 420 respectively. Enter the beginning...
-
Wyandotte Chemical Company sells various chemicals to the automobile industry. Wyandotte currently sells 30,000 gallons of polyol per year at an average price of $15 per gallon. Fixed costs of...
-
11.A simple LR circuit is connected to a battery at t = 0. The time instant at which rate of energy storage in inductor is half of power delivered by battery 2L (1) In 2 (3) In 2 (2) In (4) (4) In 3
-
Compute a depth-two decision tree for the training data in table 1 using the Gini function, C(a) = 2a(1 a) as described in class. What is the overall accuracy on the training data of the tree? XYZ...
-
Using the Net Operating Income / Cap Rate = Value formula, given the following information, calculate the overall capitalization rate. Sales price: $1,200,000. Potential Gross Income: $250,000....
-
Which of the following statements concerning the reporting of general long-term liabilities is true? a. General long-term liabilities are reported both in the governmental funds and the...
-
The Balance Sheet for governmental funds would potentially include all of the following items except a. liens receivable. b. deferred revenue. c. bonds payable due in 5 years. d. assigned fund...
-
The General Fund of the village of Oxendine transferred $150,000 to a Special Revenue Fund; $35,000 to a Capital Projects Fund; and $25,000 to Enterprise Fund A. In the same reporting period,...
-
Assume that a wealthy alumnus donated $1 million to Chavis University to provide loans to qualifying students. Though not required by the donor, the universitys Board of Trustees voted to supplement...
-
The General Fund of the village of Oxendine transferred $150,000 to a Special Revenue Fund; $35,000 to a Capital Projects Fund; and $25,000 to Enterprise Fund A. In the same reporting period,...
-
What are accrued liabilities? How do they arise? Explain. What is stored value card liability and current portion of deferred revenue? Explain each and indicate how these are related to the concept...
-
Identify one local business that uses a perpetual inventory system and another that uses a periodic system. Interview an individual in each organization who is familiar with the inventory system and...
-
Consider the cell Fe(s) FeSO 4 (aq, a = 0.0250) Hg 2 SO 4 (s) Hg(l). a. Write the cell reaction. b. Calculate the cell potential, the equilibrium constant for the cell reaction, and G o R at 25C.
-
Why can we conclude that the wave function (x, t) = (x) e i (E / h )t represents a standing wave?
-
Why was the following qualification made in Section 3.7? Note that Equation (3.47) is only applicable to a process in which there is no change in the phase of the system, such as vaporization or...
-
For the given periodic process execution times and periods (P1 has the highest priority), show how much CPU time of higher-priority processes will be required during one period of each of the...
-
For the periodic processes shown below: a. Schedule the processes using an RMS policy. b. Schedule the processes using an EDF policy. In each case, compute the schedule for an interval equal to the...
-
For the given set of periodic processes, all of which share the same deadline of 12: a. Schedule the processes for the given arrival times using standard ratemonotonic scheduling (no data...

Study smarter with the SolutionInn App



