The volume occupied by an atom is greater than the volume of the nucleus by a...
Fantastic news! We've Found the answer you've been seeking!
Question:
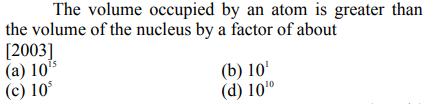
Transcribed Image Text:
The volume occupied by an atom is greater than the volume of the nucleus by a factor of about [2003] (a) 105 (c) 10 (b) 10 (d) 100 The volume occupied by an atom is greater than the volume of the nucleus by a factor of about [2003] (a) 105 (c) 10 (b) 10 (d) 100
Expert Answer:
Related Book For 

Thermodynamics for Engineers
ISBN: ?978-1133112860
1st edition
Authors: Kenneth A. Kroos, Merle C. Potter
Posted Date:
Students also viewed these physics questions
-
The volume occupied by a gas depends linearly on degrees Celsius at constant pressure, but it is not directly proportional to degrees Celsius. However, it is directly proportional to kelvins. What is...
-
The volume occupied by 20 kg of water at 300 C and 10 MPa is nearest: (A) 0.028 m3 (B) 0.088 m3 (C) 0.22 m3 (D) 0.36 m3
-
The volume occupied by one gram-equivalent of oxygen at 273 K and 550 mmHg is A) 7.74 L B) 5.6 L C) 6.84 L D) 6.08 L
-
Briar Company manufactures and sells dresses at a variable cost of $32 each and a fixed cost of x. It can sell 6,600 dresses at a selling price of $60 to earn an operating income of $14,800 (Option...
-
Rigsby Resins Company processes a base chemical into plastic. Standard costs and actual costs for direct materials, direct labor, and factory overhead incurred for the manufacture of 2,700 units of...
-
A partial alphabetized list of adjusted account balances is shown for Dover Pacific Tours as at November 30, 2020 (all accounts have normal balances). Pat Dover, the owner, uses the following account...
-
A stock has volatility \(\sigma=.30\) and a current value of \(\$ 36\). An American put option on this stock has a strike price of \(\$ 40\), and expiration is in 5 months. The interest rate is \(8...
-
A TiW layer is deposited on a substrate using a sputtering tool. Table 6E.14 contains layer thickness measurements (in angstroms) on 20 subgroups of four substrates. (a) Setup x and R control charts...
-
At December 31, 2022, Palestine Commercial Bank reports the following balance sheet information: Current assets $800,000 Noncurrent assets 2,600,000 Goodwill 300,000 Current liabilities (700,000)...
-
John Parsons (123-45-6781) and George Smith (123-45-6782) are 70% and 30% owners, respectively, of Premium, Inc. (11-1111111), a candy company located at 1005 16th Street, Cut and Shoot, TX 77303....
-
19. Write the balanced equation for the following reactions i. Substitution reaction between methane and chlorine ii. Esterification reaction between ethanoic acid and ethanol iii. Addition reaction...
-
What aspects of population policy alternatives including their strengths and weaknessesare illustrated by the cases of China and India?
-
Explain the differences between characteristics of public goods and private goods and services. How can goods and services provided by nongovernmental organisations be considered in this framework?
-
If a reform improves everyones income on average, why might people vote against it? You may wish to provide one or more numerical examples to illustrate your answer.
-
What can be learned from the historical experiences of Costa Rica and Guatemala about internal conflicts, and about both positive and negative foreign influences?
-
What lessons can be learned for low-income countries from Botswanas successes?
-
If 3000 dollars is invested in a bank account at an interest rate of 6 per cent per year, Find the amount in the bank after 5 years if interest is compounded annually: Find the amount in the bank...
-
If the joint cost function for two products is C(x, y) = xy2 + 1 dollars (a) Find the marginal cost (function) with respect to x. (b) Find the marginal cost with respect to y.
-
Air is heated from 20 C to 800 C at constant pressure of 200 kPa in a cylinder with an initial volume of 4000 cm3. The entropy change, assuming an ideal gas with constant specific heats, is nearest:...
-
Superheated steam enters an insulated turbine at 20 MPa and 600 C at a mass flow rate of 1000 kg/min. If the steam leaves the turbine at a) 10 kPa, b) 40 kPa, c) 80 kPa as a saturated vapor, what are...
-
Air at 700C is expanded in an insulated cylinder such that the volume increases by a factor of 8. Estimate the final temperature, assuming a quasi-equilibrium process. Also, calculate the work...
-
Gymnastics4Life (G4L) is a high-end facility for beginning, intermediate, and elite gymnasts. The latter are drawn from the nearby region for exclusive and dedicated training. In order to maintain...
-
CustomMetalworks is considering expanding their cable fabrication business for towers, rigging, winches, and many other uses. They have available \(\$ 250,000\) for investment and have identified the...
-
Sebastian is about to compare a set of mutually exclusive and indivisible alternatives using a ranking approach. Which of the following is not an appropriate measure of worth? a. Present worth b....

Study smarter with the SolutionInn App



