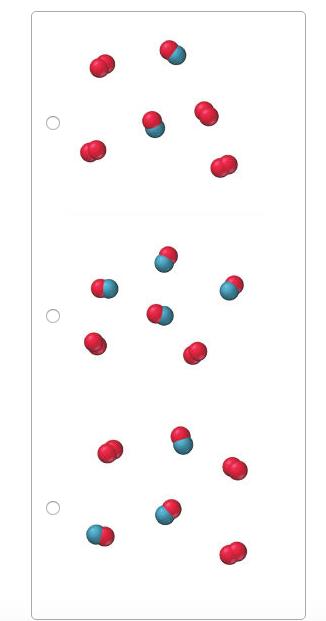
This reaction was experimentally determined to be first order with respect to O2 and second order with
Fantastic news! We've Found the answer you've been seeking!
Question:
This reaction was experimentally determined to be first order with respect to O2 and second order with respect to NO. O2(g)+2NO(g)?2NO2(g) These diagrams represent reaction mixtures in which the number of each type of molecule represents its relative initial concentration.
Which mixture has the fastest initial rate?
Related Book For 

Posted Date:




