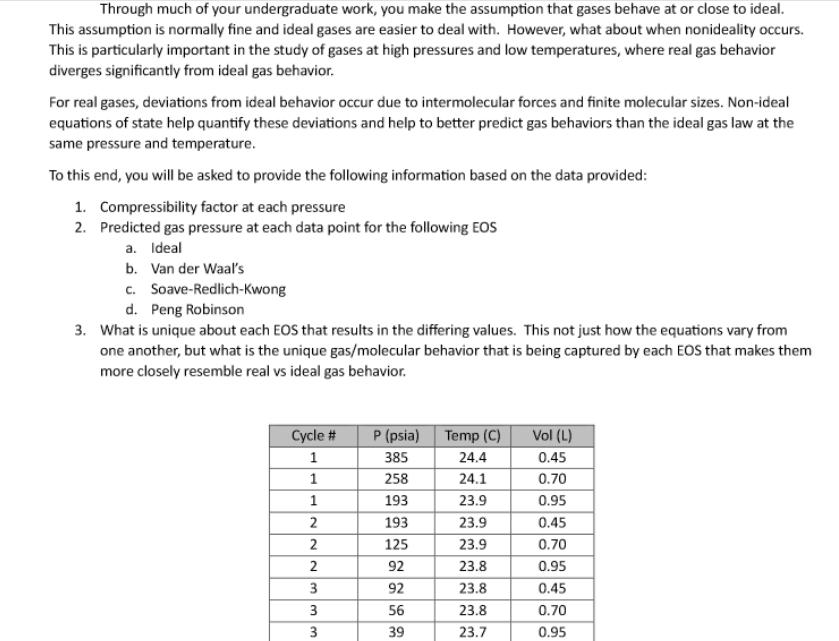
Through much of your undergraduate work, you make the assumption that gases behave at or close...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
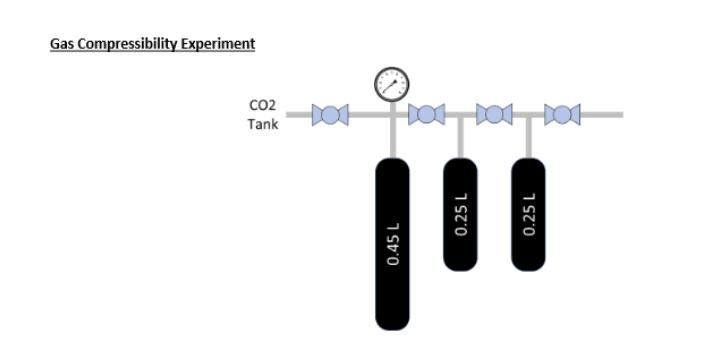
Through much of your undergraduate work, you make the assumption that gases behave at or close to ideal. This assumption is normally fine and ideal gases are easier to deal with. However, what about when nonideality occurs. This is particularly important in the study of gases at high pressures and low temperatures, where real gas behavior diverges significantly from ideal gas behavior. For real gases, deviations from ideal behavior occur due to intermolecular forces and finite molecular sizes. Non-ideal equations of state help quantify these deviations and help to better predict gas behaviors than the ideal gas law at the same pressure and temperature. To this end, you will be asked to provide the following information based on the data provided: 1. Compressibility factor at each pressure 2. Predicted gas pressure at each data point for the following EOS a. Ideal b. Van der Waal's c. Soave-Redlich-Kwong d. Peng Robinson 3. What is unique about each EOS that results in the differing values. This not just how the equations vary from one another, but what is the unique gas/molecular behavior that is being captured by each EOS that makes them more closely resemble real vs ideal gas behavior. Cycle # P (psia) Temp (C) Vol (L) 1 385 24.4 0.45 1 258 24.1 0.70 1 193 23.9 0.95 2 193 23.9 0.45 2 125 23.9 0.70 2 92 23.8 0.95 3 92 23.8 0.45 3 56 23.8 0.70 3 39 23.7 0.95 Gas Compressibility Experiment 0.45 L CO2 Tank 101 101 101 0.25 L 0.25 L Through much of your undergraduate work, you make the assumption that gases behave at or close to ideal. This assumption is normally fine and ideal gases are easier to deal with. However, what about when nonideality occurs. This is particularly important in the study of gases at high pressures and low temperatures, where real gas behavior diverges significantly from ideal gas behavior. For real gases, deviations from ideal behavior occur due to intermolecular forces and finite molecular sizes. Non-ideal equations of state help quantify these deviations and help to better predict gas behaviors than the ideal gas law at the same pressure and temperature. To this end, you will be asked to provide the following information based on the data provided: 1. Compressibility factor at each pressure 2. Predicted gas pressure at each data point for the following EOS a. Ideal b. Van der Waal's c. Soave-Redlich-Kwong d. Peng Robinson 3. What is unique about each EOS that results in the differing values. This not just how the equations vary from one another, but what is the unique gas/molecular behavior that is being captured by each EOS that makes them more closely resemble real vs ideal gas behavior. Cycle # P (psia) Temp (C) Vol (L) 1 385 24.4 0.45 1 258 24.1 0.70 1 193 23.9 0.95 2 193 23.9 0.45 2 125 23.9 0.70 2 92 23.8 0.95 3 92 23.8 0.45 3 56 23.8 0.70 3 39 23.7 0.95 Gas Compressibility Experiment 0.45 L CO2 Tank 101 101 101 0.25 L 0.25 L
Expert Answer:

Related Book For 

Fundamentals of Physics
ISBN: 978-1118230725
10th Extended edition
Authors: Jearl Walker, Halliday Resnick
Posted Date:
Students also viewed these chemical engineering questions
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
The root cause of underdevelopment and environmental degradation is the overdevelopment of a handful of rich nations. Discuss.
-
Suppose that the Treasury bill rate is 6% rather than 2%. Assume that the expected return on the market stays at 9%. Use the betas in Table 8.2. a. Calculate the expected return from Johnson &...
-
Let I n = x n e -x2 dx, where n is a nonnegative integer. a. I 0 = e -x2 dx cannot be expressed in terms of elementary functions. Evaluate I 1 . b. Use integration by parts to evaluate I 3 . c. Use...
-
A snow-cone machine at an icecream shop costs \(\$ 15,000\). The machine is expected to generate profits of \(\$ 2,500\) each year of its 10-year useful life. At the end of the 10 years, the machine...
-
Turney Company produces and sells automobile batteries, the heavy-duty HD-240. The 2014 sales forecast is as follows. Quarter HD-240 1 ......... 5,000 2 ......... 7,000 3 ......... 8,000 4 .............
-
Let M be a metric space. If {x} and {y} are sequences in M such that x x and y y, show that d(xn, yn) d(x,y).
-
Suppose you were short in a September T-bond futures contract at 93-16. What would your profit or loss be at the September expiration if the cheapest deliverable bond you could purchase in the market...
-
The unadjusted trial balance of Simple Consulting Services as at December 31, 2021 is as follows: Cash Accounts receivable Prepaid insurance Supplies inventory Office equipment Accumulated...
-
create the post closing trial balance
-
Assume that Sally Williams desires to accumulate $1,000,000 in 15 years using her money market fund balance of $182,696. At what interest rate must Sally's investment compound annually? Interest rate...
-
Two masses are suspended by a cord that passes over a pulley with negligible mass. The cord also has negligible mass. One of the masses, m 1 , has a mass of 6.0 kg and the other mass, m2, has a mass...
-
A charged particle traveling in the y - direction with a momentum of 0 . 0 1 kg m/s enters a regions of space that has a uniform 1 Tesla magnetic. field in the z- direction as shown the Figure, If...
-
Evaluate the following numerical expressions in your head, then use the active code window to check your results: 5 ** 2 9 * 5 15 / 12 12 / 15 15 // 12 12 // 15 5 % 2 9 % 5 15 % 12 12 % 15 6 % 6 0 % 7
-
For the following exercises, rewrite the sum as a product of two functions or the product as a sum of two functions. Give your answer in terms of sines and cosines. Then evaluate the final answer...
-
Figure 40-25 is an energy-level diagram for a fictitious three-dimensional infinite potential well that contains one electron. The number of degenerate states of the levels is indicated: "non" means...
-
A positive tau ((+, rest energy = 1777 MeV) is moving with 2200 MeV of kinetic energy in a circular path perpendicular to a uniform 1.20 T magnetic field. (a) Calculate the momentum of the tau in...
-
(Distinguish carefully between atomic and nuclear masses, and take the positrons properly into account.) 1 1.007 825 4 4.002 603 2.014102 _ 0.000 548 6 3.016 029
-
Briefly describe what is meant by DEI efforts.
-
Explain each of the four examples of a bona fide occupational qualification.
-
What important precedents were set by the Griggs v. Duke Power Company case? The Albemarle v. Moody case?

Study smarter with the SolutionInn App


