Consider the ion He 2 2+? (charge on each nucleus A or B is Z=2). a) Using
Question:
Consider the ion He22+?(charge on each nucleus A or B is Z=2).
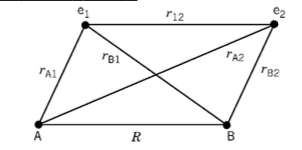
a) Using the diagram above write down the Hamiltonian for the He22+ ion explicitly including all kinetic energy and potential energy terms (Hint: it may be easier to work in atomic units)
b) He22+ is a multielectron molecular ion and, therefore, the wave function has to account for electron 2+ 2spin. The He22+ ion has electronic configuration (1?g)2 . Write down the Slater determinant for its ground state
c) Write down the Slater determinant for the ground state of the He-H+ ion. What is the same and what is different with the He22+ Slater determinant? Explain your answer.
d) Now consider adding an extra electron to He22+ to produce He2+ . Write down the Slater determinant for He2+.





