What is the goal of a regression model/equation? From the beginning, describe the steps you would...
Fantastic news! We've Found the answer you've been seeking!
Question:
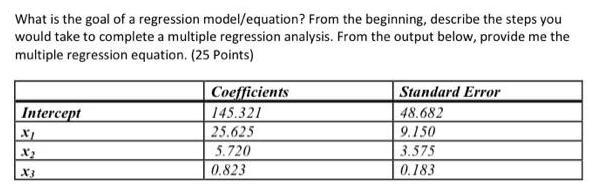
Transcribed Image Text:
What is the goal of a regression model/equation? From the beginning, describe the steps you would take to complete a multiple regression analysis. From the output below, provide me the multiple regression equation. (25 Points) Intercept X₁ X3 Coefficients 145.321 25.625 5.720 0.823 Standard Error 48.682 9.150 3.575 0.183 What is the goal of a regression model/equation? From the beginning, describe the steps you would take to complete a multiple regression analysis. From the output below, provide me the multiple regression equation. (25 Points) Intercept X₁ X3 Coefficients 145.321 25.625 5.720 0.823 Standard Error 48.682 9.150 3.575 0.183
Expert Answer:
Answer rating: 100% (QA)
Based on the output provided the multiple regression equation is Y 145321 25625 X 5720 X2 0823 X3 Th... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
The Arnold Company has the following information relating to its inventory for 2009 Beginning inventory 80000 Ending inventory 60000 REQUIRED Create the entries under each of the two systems that...
-
The Webster Store shows the following information relating to one of its products. Inventory, January 1 . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .250 units @ $17.00...
-
Nike, Inc., reports the following information relating to its manufacturing activities in the footnotes to its 2018 10-K report: We are supplied by 124 footwear factories located in 13 countries. The...
-
Suppose that a 7% semi-annual coupon bond with a time to maturity of 8 years and a par value of $100 has a price of $106,4. This bond is first callable in 6 years at a redemption price of $104,8....
-
After adjusting entries had been made, Infosys Companys balance sheets showed the following asset and liability amounts at the end of 2013 and 2014: The following amounts were taken from the 2014...
-
A work area has a reflectivity of 60 percent, based on the color combinations of the workstations and the immediate environment. The seeing task of the assembly work could be classified as difficult....
-
These charged particles are traveling in circular orbits with velocities and field directions as noted. Which particles have a negative charge? x X x A. B. C. D.
-
Sudss Bottling Company does bottling Labeling, and distribution work for several local microbreweries. The demand rate for Wortmans beer is 600 cases [24 bottles each) per week. Sudss bottling...
-
Why would a company simultaneously sell the building to a company and at the same time lease it back from this same company for 20 years?
-
Complete the missing amounts in the following production report. Materials are added at the beginning of the process; conversion costs are incurred evenly; the ending inventory is 60% complete. The...
-
A put-on gold is struck at $1900. What happens if you exercise when the futures price is $1877 and the futures contract settles at $1879? How much money will you make? (Hint: there are two possible...
-
Technician A says an ignition scope can be used to diagnose a stretched timing chain. Technician B says timing chain stretch can be diagnosed by observing distributor rotor movement when the...
-
Draw a Context diagram and a Data Flow Diagram for a manufacturing company with a material handling and financial system that works as follows: Widget Corporation makes high-tech fly swatters. The...
-
Discuss the loss relief available to a both an individual and a trading company in the UK ?
-
You are considering investing $100,000 in a project that is to generate dividends of $10,000 in one year. You expect that dividends will grow at 2% per year for the foreseeable future and the rate of...
-
Write essay on my first date
-
For each of the engineering requirements below, determine if it meets the proper- ties of abstractness, verifiable, unambiguous, and realistic. If a requirement does not satisfy the properties,...
-
Chris Zulliger was a chef at the Plaza Restaurant in the Snowbird Ski Resort in Utah. The restaurant is located at the base of a mountain. As a chef for the Plaza, Zulliger was instructed by his...
-
The atomic masses of 63Li and 73Li are 6.0151 amu and 7.0160 amu, respectively. Calculate the natural abundances of these two isotopes. The average atomic mass of Li is 6.941 amu.
-
(a) The free Cu(I) ion is unstable in solution and has a tendency to disproportionate: Use the information in Table 19.1 to calculate the equilibrium constant for the reaction. (b) Based on your...
-
Determine the standard enthalpy of formation of ethanol (C2H5OH) from its standard enthalpy of combustion (-1367.4 kJ/mol)?
-
In the FRED database, find a variable that is available in both a seasonally adjusted form and a not seasonally adjusted form. Plot both over time and describe how large the seasonal variation in the...
-
Graph the levels of real GDP for the United States, Canada, and Germany (data can be found at www.oecd.org under Statistics and then under National Accounts). Are U.S. and Canadian business cycles...
-
How does each of the following variables behave over the business cycle? Develop graphs to show your results and give economic explanations. a. Real imports b. Federal government receipts c. Housing...

Study smarter with the SolutionInn App


