Titration of a 50.00 ml sample of acid rain required 9.30 mL of 0.0012 M NaOH to
Fantastic news! We've Found the answer you've been seeking!
Question:
Titration of a 50.00 ml sample of acid rain required 9.30 mL of 0.0012 M NaOH to reach the end point. What was the total [H3O+] in the rain sample? What was the pH? (see Chemistry in Action: Acid Rain).
Chemistry In Action
Acid Rain


![]()

Step-by-step?
Transcribed Image Text:
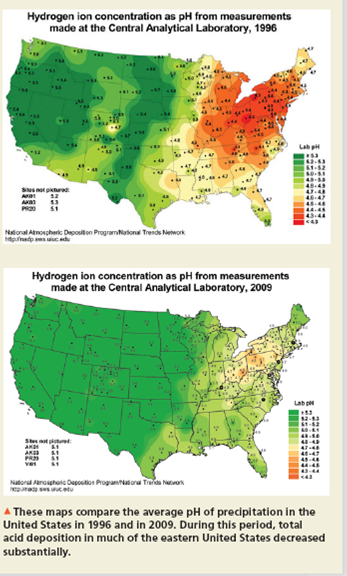
As the water that evaporates from oceans and lakes condenses into raindrops, it dissolves small quantities of gases from the atmosphere. Under normal conditions, rain is slightly acidic, with a pH close to 5.6, because of atmospheric CO₂ that dis- solves to form carbonic acid: COz(aq) + H2O(0) = H₂CO₂(aq) → HCO₂ (aq) + H₂0+ (aq) In recent decades, however, the acidity of rainwater in many industrialized areas of the world has increased by a factor of over 100, to a pH between 3 and 3.5. The primary cause of this so-called acid rain is industrial and automotive pollution. Each year, large power plants and smelt- ers pour millions of tons of sulfur dioxide (SO₂) gas into the atmosphere, where some is oxidized by air to produce sulfur tri- oxide (SO₂). Sulfur oxides then dissolve in rain to form dilute sulfurous acid (H₂SO₂) and sulfuric acid (H₂SO₂): SO₂(g) + H₂O(l) →→→ H₂SO₂ (aq) SO₂(g) + H₂O(1) H₂SO₂ (aq) Nitrogen oxides produced by the high-temperature reaction of N₂ with O₂ in coal-burning plants and in automobile engines fur- ther contribute to the problem. Nitrogen dioxide (NO₂) dissolves in water to form dilute nitric acid (HNO3) and nitric oxide (NO): 3 NO₂(g) + H₂O() →→→ 2 HNO₂ (aq) + NO(g) Oxides of both sulfur and nitrogen have always been pres- ent in the atmosphere, produced by such natural sources as vol- canoes and lightning bolts, but their amounts have increased dramatically over the last century because of industrialization. The result is a notable decrease in the pH of rainwater in more densely populated regions, including Europe and the eastern United States. Many processes in nature require such a fine pH balance that they are dramatically upset by the shift that has occurred in the pH of rain. Some watersheds contain soils that have high "buffering capacity" and so are able to neutralize acidic compounds in acid rain. Other areas, such as the northeastern United States and eastern Canada, where soil-buffering capac- ity is poor, have experienced negative ecological effects. Acid rain releases aluminum salts from soil, and the ions then wash into streams. The low pH and increased aluminum levels are so toxic to fish and other organisms that many lakes and streams in these areas are devoid of aquatic life. Massive tree die-offs have occurred throughout central and eastern Europe as acid rain has lowered the pH of the soil and has leached nutrients from leaves. Fortunately, adidic emissions in the United States have been greatly reduced in recent years as a result of the Clean Air Act Amendments of 1990. Industrial emissions of SO₂ and nitrogen oxides decreased by over 40% from 1990 to 2007, resulting in a decrease in acid rain depositions, particularly in the eastern United States and Canada (see accompanying figure). While significant reductions have been realized, most environmental scientists agree that additional reductions in these pollutant emissions are necessary to ensure the recovery of affected lakes and streams. This limestone statue adorning the Rheims Cathedral in France has been severely eroded by acid rain. Hydrogen ion concentration as pH from measurements made at the Central Analytical Laboratory, 1996 Sus not pr sk St HAT E suced -AP 54 .. National Deposition Program Nasional Tr 1 National Atmospheric Deposition Program National Trends Network +48 20 +47 la ** Hydrogen ion concentration as pH from measurements made at the Central Analytical Laboratory, 2009 7.0 1.4. y4 Lab ▲ These maps compare the average pH of precipitation in the United States in 1996 and in 2009. During this period, total acid deposition in much of the eastern United States decreased substantially. As the water that evaporates from oceans and lakes condenses into raindrops, it dissolves small quantities of gases from the atmosphere. Under normal conditions, rain is slightly acidic, with a pH close to 5.6, because of atmospheric CO₂ that dis- solves to form carbonic acid: COz(aq) + H2O(0) = H₂CO₂(aq) → HCO₂ (aq) + H₂0+ (aq) In recent decades, however, the acidity of rainwater in many industrialized areas of the world has increased by a factor of over 100, to a pH between 3 and 3.5. The primary cause of this so-called acid rain is industrial and automotive pollution. Each year, large power plants and smelt- ers pour millions of tons of sulfur dioxide (SO₂) gas into the atmosphere, where some is oxidized by air to produce sulfur tri- oxide (SO₂). Sulfur oxides then dissolve in rain to form dilute sulfurous acid (H₂SO₂) and sulfuric acid (H₂SO₂): SO₂(g) + H₂O(l) →→→ H₂SO₂ (aq) SO₂(g) + H₂O(1) H₂SO₂ (aq) Nitrogen oxides produced by the high-temperature reaction of N₂ with O₂ in coal-burning plants and in automobile engines fur- ther contribute to the problem. Nitrogen dioxide (NO₂) dissolves in water to form dilute nitric acid (HNO3) and nitric oxide (NO): 3 NO₂(g) + H₂O() →→→ 2 HNO₂ (aq) + NO(g) Oxides of both sulfur and nitrogen have always been pres- ent in the atmosphere, produced by such natural sources as vol- canoes and lightning bolts, but their amounts have increased dramatically over the last century because of industrialization. The result is a notable decrease in the pH of rainwater in more densely populated regions, including Europe and the eastern United States. Many processes in nature require such a fine pH balance that they are dramatically upset by the shift that has occurred in the pH of rain. Some watersheds contain soils that have high "buffering capacity" and so are able to neutralize acidic compounds in acid rain. Other areas, such as the northeastern United States and eastern Canada, where soil-buffering capac- ity is poor, have experienced negative ecological effects. Acid rain releases aluminum salts from soil, and the ions then wash into streams. The low pH and increased aluminum levels are so toxic to fish and other organisms that many lakes and streams in these areas are devoid of aquatic life. Massive tree die-offs have occurred throughout central and eastern Europe as acid rain has lowered the pH of the soil and has leached nutrients from leaves. Fortunately, adidic emissions in the United States have been greatly reduced in recent years as a result of the Clean Air Act Amendments of 1990. Industrial emissions of SO₂ and nitrogen oxides decreased by over 40% from 1990 to 2007, resulting in a decrease in acid rain depositions, particularly in the eastern United States and Canada (see accompanying figure). While significant reductions have been realized, most environmental scientists agree that additional reductions in these pollutant emissions are necessary to ensure the recovery of affected lakes and streams. This limestone statue adorning the Rheims Cathedral in France has been severely eroded by acid rain. Hydrogen ion concentration as pH from measurements made at the Central Analytical Laboratory, 1996 Sus not pr sk St HAT E suced -AP 54 .. National Deposition Program Nasional Tr 1 National Atmospheric Deposition Program National Trends Network +48 20 +47 la ** Hydrogen ion concentration as pH from measurements made at the Central Analytical Laboratory, 2009 7.0 1.4. y4 Lab ▲ These maps compare the average pH of precipitation in the United States in 1996 and in 2009. During this period, total acid deposition in much of the eastern United States decreased substantially.
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
In a titration experiment, 12.5 mL of 0.500 M H2SO4 neutralize 50.0 mL of NaOH. What is the concentration of the NaOH solution?
-
In a titration experiment, 20.4 mL of 0.883 M HCOOH neutralize 19.3 mL of Ba(OH)2. What is the concentration of the Ba(OH)2 solution?
-
A 3.0 m long beam has a cross-sectional area of 0.012 m2. It is subjected to a tensile force of 2.0 kN. If its modulus of elasticity is 300 MPa, how much does the beam stretch in mm approximately?
-
Julia Robertson is a senior at Tech, and she's investigating different ways to finance her final year at school. She is considering leasing a food booth outside the Tech stadium at home football...
-
Calculate [y] for the two-port in Fig. 19.85. 4 2
-
1. Use methods of descriptive statistics to summarize the data. 2. Develop an estimated simple linear regression model that can be used to predict the alumni giving rate, given the graduation rate....
-
Indicate the type of flow given by \(\phi=\sqrt{r} \cos (\theta / 2)\). Calculate and plot typical streamlines.
-
The American Express Company is a major financial services company, noted for its American Express card. Below are some of the performance measures used by the company in its balanced scorecard....
-
Table below shows the demand for haircuts from seniors and other customers on an average weekday in the local hairdressing shop. Price of Haircut Quantity Demanded by Seniors 2 Quantity Demanded by...
-
Emergency calls to the 911 system of Durham, North Carolina, for the past 24 weeks are shown in the following table: a) Compute the exponentially smoothed forecast of calls for each week. Assume an...
-
The combination of weakening oil prices and the failure of Congress to deal with the budget deficit by cutting spending led some to see the possibility of achieving two objectives at once: (1)...
-
What is gliding locomotion?
-
Who falls faster when wearing the same-size parachutea heavy person or a light personor do both fall at the same speed?
-
Describe some of the physical characteristics that gliding organisms have evolved to increase their surface area.
-
How does the vertical component of motion for a ball kicked off a high cliff compare with the motion of vertical free fall?
-
What is fund accounting?
-
In the process of saving some money for my kids college I plan to make six annual deposits of $4000 into a secret savings account that pays an interest of 4% compounded annually. Two years after...
-
You are a U.S. investor who purchased British securities for 2,000 one year ago when the British pound cost U.S. $1.50. What is your total return (based on U.S. dollars) if the value of the...
-
Discuss the following phenomena in terms of the gas laws: (a) The pressure increase in an automobile tire on a hot day. (b) The "popping" of a paper bag. (c) The expansion of a weather balloon as it...
-
Draw the Lewis structure for the BeCl42- ion. Predict its geometry and describe the hybridization state of the Be atom?
-
Referring to Table 10.3, arrange the following molecules in order of increasing dipole moment: H2O, H2S, H2Te, H2Se?
-
Differentiate between tax compliance and tax planning.
-
Differentiate between an open transaction and a closed transaction.
-
What form(s) is (are) used to file an individuals income tax return?

Study smarter with the SolutionInn App


