TLC of Known Compounds Complete the table below (include calculations, use additional page as necessary) Compound...
Fantastic news! We've Found the answer you've been seeking!
Question:

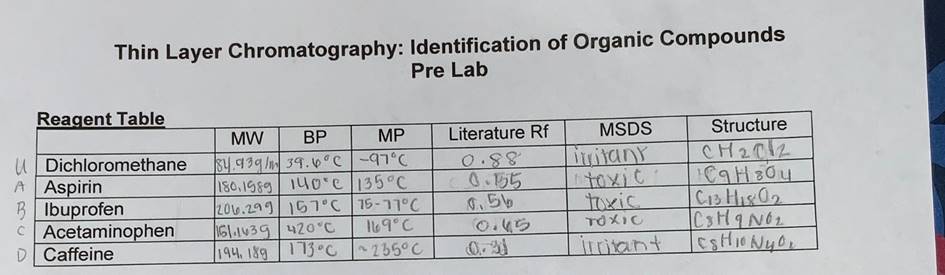

Transcribed Image Text:
TLC of Known Compounds Complete the table below (include calculations, use additional page as necessary) Compound Experimental Rf Aspirin Ibuprofen Acetaminophen Caffeine 0.60 0.58 0.625 0.375 Unknown ID A Experimental Rf 0.625 RF distance traveled by solute Aspirin = 3.0 =0.60 5.0 TLC of Unknown Compounds Caffeine = 1.5 4.0 = 0.375 2. 15pts Complete the table below (include calculations, use additional page as necessary). = 0.625 distance traveled by solven + Ibu. = 2.9 = 0,58 50 unk. 9 Acetaminophen = 2.5 = 0.625 4.0 2,5 4.0 3. 5pts What is the identity of your unknown? Briefly describe all of the data you have to support your identification. of The identity unknown was acetaminophen. I know this was my unknown because in my my picture of The second TLC plate, the spot where letter "C" (acelaminophen was even with the spot of my unknown. A. 4. 20 Do the experimental Rf values match the predicted rate of elution based on the chemical/structural properties of each molecule? Why or Why not? Include structures and describe any deviations. For each molecule the rf values I calculated were very close to the literature values. Thin Layer Chromatography: Identification of Organic Compounds Pre Lab Reagent Table UDichloromethane A Aspirin BIbuprofen Acetaminophen D Caffeine MW MP BP 84.939/1 39.6°C -97°C 180.1989 140°C 135°C 206.299 157°C 15-17°C 169°C 161.1439 420°C 194, 189 173°C ~235°C Literature Rf 0.88 0.155 0.56 0.31 MSDS Limitany toxic TOXIC irritant Structure CH ₂012 C9H304 C13H180₂ Cs Ha Noi |CSHIONYO₂ BIOMASS BOILER OPERATION ANALYSIS A fuel silo stores the biomass waste fuel and is fed into the boiler using a conveyor system. Combustion air is preheated before entering the boiler using an air preheater system that regenerates waste heat from the boiler flue gas. Due to the high temperature difference between the flue gas and ambient, the air preheater operates under non-adiabatic where a 10% heat loss is expected from its adiabatic heat transfer rate from the gas to the combustion air. QUESTION 1 [CO1] Select a suitable biomass waste fuel and obtain its volumetric composition from available literature sources. QUESTION 2 [CO2] The flue gas acts as a heating source for the combustion air. The flue gas inlet temperature and flow rate into the air preheater are constant at 250°C and 100 litres/min, while the air inlet temperature and flow rate into the air preheater are constant at 30°C and 40 litres/min. 2.1) Obtain from literature sources the flue gas density from the combustion process of the selected biomass fuel. 2.2) Based on the sensible heating analysis of air, plot the flue gas exit (exhaust) temperature when the preheated combustion air exits the preheater between 40°C and 70°C. 2.3) Then, calculate the maximum waste heat recovery percentage from the flue gas for this air preheating process, based on Eq. (1). Waste heat recovery ratio, EWHR Qgas-air Qgas (1) QUESTION 3 [CO3] If the supplied excess air is fixed at 130%, plot the combustion heat transfer rate (Qc) profile from the combustion of the selected biomass fuel as the combustion air temperature changes from 40°C and 70°C. TLC of Known Compounds Complete the table below (include calculations, use additional page as necessary) Compound Experimental Rf Aspirin Ibuprofen Acetaminophen Caffeine 0.60 0.58 0.625 0.375 Unknown ID A Experimental Rf 0.625 RF distance traveled by solute Aspirin = 3.0 =0.60 5.0 TLC of Unknown Compounds Caffeine = 1.5 4.0 = 0.375 2. 15pts Complete the table below (include calculations, use additional page as necessary). = 0.625 distance traveled by solven + Ibu. = 2.9 = 0,58 50 unk. 9 Acetaminophen = 2.5 = 0.625 4.0 2,5 4.0 3. 5pts What is the identity of your unknown? Briefly describe all of the data you have to support your identification. of The identity unknown was acetaminophen. I know this was my unknown because in my my picture of The second TLC plate, the spot where letter "C" (acelaminophen was even with the spot of my unknown. A. 4. 20 Do the experimental Rf values match the predicted rate of elution based on the chemical/structural properties of each molecule? Why or Why not? Include structures and describe any deviations. For each molecule the rf values I calculated were very close to the literature values. Thin Layer Chromatography: Identification of Organic Compounds Pre Lab Reagent Table UDichloromethane A Aspirin BIbuprofen Acetaminophen D Caffeine MW MP BP 84.939/1 39.6°C -97°C 180.1989 140°C 135°C 206.299 157°C 15-17°C 169°C 161.1439 420°C 194, 189 173°C ~235°C Literature Rf 0.88 0.155 0.56 0.31 MSDS Limitany toxic TOXIC irritant Structure CH ₂012 C9H304 C13H180₂ Cs Ha Noi |CSHIONYO₂ BIOMASS BOILER OPERATION ANALYSIS A fuel silo stores the biomass waste fuel and is fed into the boiler using a conveyor system. Combustion air is preheated before entering the boiler using an air preheater system that regenerates waste heat from the boiler flue gas. Due to the high temperature difference between the flue gas and ambient, the air preheater operates under non-adiabatic where a 10% heat loss is expected from its adiabatic heat transfer rate from the gas to the combustion air. QUESTION 1 [CO1] Select a suitable biomass waste fuel and obtain its volumetric composition from available literature sources. QUESTION 2 [CO2] The flue gas acts as a heating source for the combustion air. The flue gas inlet temperature and flow rate into the air preheater are constant at 250°C and 100 litres/min, while the air inlet temperature and flow rate into the air preheater are constant at 30°C and 40 litres/min. 2.1) Obtain from literature sources the flue gas density from the combustion process of the selected biomass fuel. 2.2) Based on the sensible heating analysis of air, plot the flue gas exit (exhaust) temperature when the preheated combustion air exits the preheater between 40°C and 70°C. 2.3) Then, calculate the maximum waste heat recovery percentage from the flue gas for this air preheating process, based on Eq. (1). Waste heat recovery ratio, EWHR Qgas-air Qgas (1) QUESTION 3 [CO3] If the supplied excess air is fixed at 130%, plot the combustion heat transfer rate (Qc) profile from the combustion of the selected biomass fuel as the combustion air temperature changes from 40°C and 70°C.
Expert Answer:
Answer rating: 100% (QA)
TLC Analysis of Unknown Based on the information provided your unknown compound has an experimental ... View the full answer

Related Book For 

Horngrens Financial and Managerial Accounting
ISBN: 978-0133866292
5th edition
Authors: Tracie L. Nobles, Brenda L. Mattison, Ella Mae Matsumura
Posted Date:
Students also viewed these accounting questions
-
Complete the table below for contribution margin per unit, total contribution margin, and contribution margin ratio: Number of units Sales price per unit Variable costs per unit Calculate:...
-
Complete the table below for contribution margin per unit, total contribution margin , and contribution margin ratio: A Number of units 1,720 units 14,920 units 4,620 units Sales price per unit $...
-
Complete the table below for contribution margin per unit, total contribution margin, and contribution marginratio: 1,300 units 100 3,600 units 40 7,500 units $125 Number of units Sales price per...
-
In Exercises find the derivative of the algebraic function. f(x) = (2x + 5x)(x 3)(x + 2)
-
Rasmussen, CPAs, completed 1,800 tax returns for clients during 2008. The average price paid per return was $ 165. The firms master budget indicated expected tax return revenues of $ 308,000 based on...
-
Just as Joe Spumonti wanted to promote one of his employees to the COO role, none seemed interested. What could Joe have done to arrive at this time with a choice between inside candidates and this...
-
The turbine shown in Fig. P8.85 develops 400 kW. Determine the flowrate if (a) head losses are negligible (b) head loss due to friction in the pipe is considered. Assume \(f=0.02\). There may be more...
-
Alyson Clark is a licensed incorporated CPA. During the first month of operations of the business, the following events and transactions occurred: Sept. 1...Invested $40,000 cash in exchange for...
-
What was the purpose for here I stand the BIOGRAPHY OF Martin Luther. Why is it important to establish the meaning of talent and talent management in a particular organizational setting? How does the...
-
Claude Lopez is the president of Zebra Antiques. His employee, Dwight Francis, is due a raise. Dwight?s current benefits analysis is as follows: Company Cost (Current) Yearly Benefit Costs Employee...
-
How does temperature distribution in deep wells differ from that in shallow wells?
-
Provide suitable hashCode and equals methods for the Pair class of Section 18.2 and implement a HashMap class, using a HashSet .
-
Write a chat server and client program. The chat server accepts connections from clients. Whenever one of the clients sends a chat message, it is displayed for all other clients to see. Use a...
-
Design a set of database tables to store library books and patrons. A book has an ISBN (International Standard Book Number), an author, and a title. The library may have multiple copies of each book,...
-
Implement a client-server program in which the client will print the date and time given by the server. Two classes should be implemented: DateClient and DateServer. The DateServer simply prints new...
-
Try out the HEAD command of the HTTP protocol. What command did you use? What response did you get?
-
Given the following block diagram r(t) 1/m K c(t) derive the differential equation relating the output c(t) to the input r(t)
-
The liquidliquid extractor in Figure 8.1 operates at 100F and a nominal pressure of 15 psia. For the feed and solvent flows shown, determine the number of equilibrium stages to extract 99.5% of the...
-
Zoes, a national manufacturer of lawn-mowing and snow-blowing equipment, segments its business according to customer type: professional and residential. The following divisional information was...
-
Top managers of Best Video are alarmed by their operating losses. They are considering dropping the DVD product line. Company accountants have prepared the following analysis to help make this...
-
At the beginning of the year, office supplies of $800 were on hand. During the year, Rocket Air Conditioning Service paid $6,000 for more office supplies. At the end of the year, Rocket has $500 of...
-
The position of a particle undergoing simple harmonic motion is given by \(x(t)=20 \cos (8 \pi t)\), where \(x\) is in millimeters and \(t\) is in seconds. For this motion, what are the (a)...
-
Fill in the blanks to make the following statements correct. a. It is difficult to compare two or more data series when absolute numbers and different units are used. For that reason we construct...
-
Fill in the blanks to make the following statements correct. a. On a graph with Y on the vertical axis and X on the horizontal axis, the slope of a straight line is calculated as ___________. b. In...

Study smarter with the SolutionInn App


