Total Metal Contents of samples Sample ID Sample 1: Crushed CRT funnel glass Sample 2: Crushed...
Fantastic news! We've Found the answer you've been seeking!
Question:
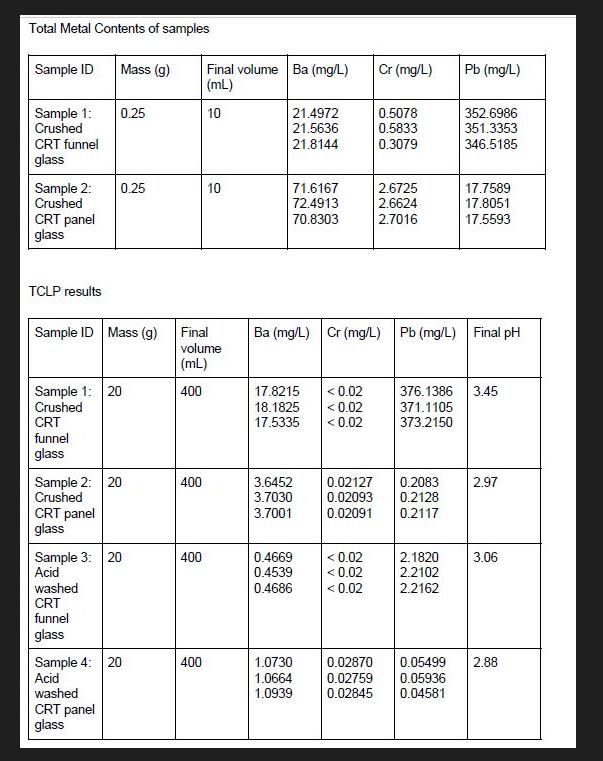
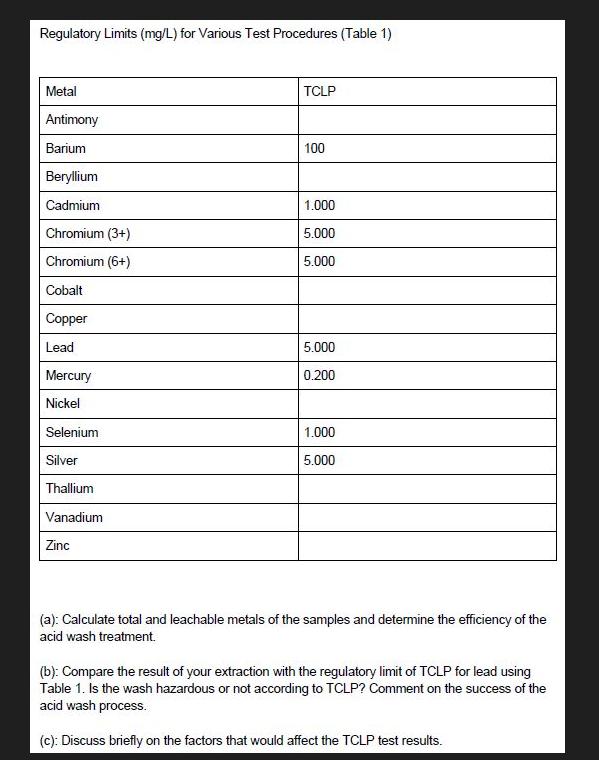
Transcribed Image Text:
Total Metal Contents of samples Sample ID Sample 1: Crushed CRT funnel glass Sample 2: Crushed CRT panel glass TCLP results CRT funnel glass Sample ID Mass (g) Mass (g) Sample 1: 20 Crushed CRT panel glass 0.25 0.25 Sample 2: 20 Crushed washed CRT funnel glass Sample 3: 20 Acid washed CRT panel glass Sample 4: 20 Acid 400 Final volume (mL) 400 400 Final volume Ba (mg/L) (mL) 10 400 10 21.4972 21.5636 21.8144 71.6167 72.4913 70.8303 17.8215 <0.02 18.1825 <0.02 17.5335 <0.02 3.6452 3.7030 3.7001 0.4669 0.4539 0.4686 1.0730 1.0664 1.0939 Cr (mg/L) Ba (mg/L) Cr (mg/L) Pb (mg/L) Final pH < 0.02 <0.02 <0.02 0.5078 0.5833 0.3079 2.6725 2.6624 2.7016 0.02870 0.02759 0.02127 0.2083 0.02093 0.2128 0.02091 0.2117 Pb (mg/L) 352.6986 351.3353 346.5185 2.1820 2.2102 2.2162 17.7589 17.8051 17.5593 376.1386 3.45 371.1105 373.2150 2.97 3.06 0.05499 2.88 0.05936 0.02845 0.04581 Regulatory Limits (mg/L) for Various Test Procedures (Table 1) Metal Antimony Barium Beryllium Cadmium Chromium (3+) Chromium (6+) Cobalt Copper Lead Mercury Nickel Selenium Silver Thallium Vanadium Zinc TCLP 100 1.000 5.000 5.000 5.000 0.200 1.000 5.000 (a): Calculate total and leachable metals of the samples and determine the efficiency of the acid wash treatment. (b): Compare the result of your extraction with the regulatory limit of TCLP for lead using Table 1. Is the wash hazardous or not according to TCLP? Comment on the success of the acid wash process. (c): Discuss briefly on the factors that would affect the TCLP test results. Total Metal Contents of samples Sample ID Sample 1: Crushed CRT funnel glass Sample 2: Crushed CRT panel glass TCLP results CRT funnel glass Sample ID Mass (g) Mass (g) Sample 1: 20 Crushed CRT panel glass 0.25 0.25 Sample 2: 20 Crushed washed CRT funnel glass Sample 3: 20 Acid washed CRT panel glass Sample 4: 20 Acid 400 Final volume (mL) 400 400 Final volume Ba (mg/L) (mL) 10 400 10 21.4972 21.5636 21.8144 71.6167 72.4913 70.8303 17.8215 <0.02 18.1825 <0.02 17.5335 <0.02 3.6452 3.7030 3.7001 0.4669 0.4539 0.4686 1.0730 1.0664 1.0939 Cr (mg/L) Ba (mg/L) Cr (mg/L) Pb (mg/L) Final pH < 0.02 <0.02 <0.02 0.5078 0.5833 0.3079 2.6725 2.6624 2.7016 0.02870 0.02759 0.02127 0.2083 0.02093 0.2128 0.02091 0.2117 Pb (mg/L) 352.6986 351.3353 346.5185 2.1820 2.2102 2.2162 17.7589 17.8051 17.5593 376.1386 3.45 371.1105 373.2150 2.97 3.06 0.05499 2.88 0.05936 0.02845 0.04581 Regulatory Limits (mg/L) for Various Test Procedures (Table 1) Metal Antimony Barium Beryllium Cadmium Chromium (3+) Chromium (6+) Cobalt Copper Lead Mercury Nickel Selenium Silver Thallium Vanadium Zinc TCLP 100 1.000 5.000 5.000 5.000 0.200 1.000 5.000 (a): Calculate total and leachable metals of the samples and determine the efficiency of the acid wash treatment. (b): Compare the result of your extraction with the regulatory limit of TCLP for lead using Table 1. Is the wash hazardous or not according to TCLP? Comment on the success of the acid wash process. (c): Discuss briefly on the factors that would affect the TCLP test results.
Expert Answer:
Answer rating: 100% (QA)
Calculate total and leachable metals of the samples and determine the efficiency of the acid wash treatment Sample 1 Crushed CRT funnel glass Total metal content Ba 3526986 mgL Cr 05833 mgL Pb 03079 m... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
(a) Calculate total and leachable metals of the samples and determine the efficiency of the acid wash treatment. (b): Compare the result of your extraction with the regulatory limit of TCLP for lead...
-
Question : 1. In the case of digital product development, how to build an ideal and successful Digital Product. Explain and give an example 2. How to define the essentials of User Interaction Design...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1 and 2. On September 1, Irene opened a retail store that specializes in sports car...
-
Walmart Stores has run into opposition when it has tried to open stores in certain towns in the United States. Walmarts capital budgeting process has determined that these locations would be...
-
Assume you own a portfolio consisting of the following stocks: a. Determine the expected return on your portfolio. b. Determine the portfolio beta (P). c. Given the portfolio beta and the assumptions...
-
Cassy Dominic, a marketing executive for Fresh Views Inc., has proposed expanding its product line of framed graphic art by producing a line of lower-quality products. These would require less...
-
Sunny Day Sunlamps, Inc., had Sales Revenue of $53 million, Sales Returns and Allowances of $2 million, and Sales Discounts of $0.3 million in 2010. Cost of goods sold was $23 million, and net income...
-
On January 1, 2010, Porter Company purchased an 80% interest in the capital stock of Salem Company for $850,000. At that time, Salem Company had capital stock of $550,000 and retained earnings of...
-
1. Calculate or estimate the following for each firm in the industry: Price/ Price/ Cash Return Projected Industry Firms Earnings Sales Flow On Equity Growth A $274 $25,313 $1,803 11.60% -12.9% B...
-
An alphabetical list of the adjusted trial balance accounts for North Country Rentals after its first year of operations ending March 31, 2020, is shown below: Required 1. Calculate the capital...
-
List and explain the main causes and consequences of global change since the 1850s to the present day.
-
You are casting high carbon steel and need a 50:1 atomic ratio of Fe:C (fifty times as many Fe atoms as C atoms) in the powder you will melt and cast. If you use 2.3 kg (2300 g) of Fe powder, what is...
-
Risk factors can lead to adversity. Protective factors can lead to resilience. We've learned that ACEs are common. So is resilience! We know that the research on ACEs is FACT, but the statistics...
-
At the surface of a steel bar there is one carbon atom per 40 unit cells of iron. At 2 mm below the surface, there is one carbon atom per 50 unit cells. The structure at 1000 C is FCC with the...
-
I would like for you to watch this video and come back to this post with your thoughts. What are the stages of changes that you have witnessed if you have already worked in the social work field?...
-
prepare the script between client and financial planner = I'm not prepared to move forward with the portfolio you've presented right now because I'm afraid that Donald Trump might take over the U.S....
-
taxpayer purchased and placed in service a computer network for his CPA practice that cost $43,000 on December 17. 2019. He did not take Section 179 expensing and opted out of bonus depreciation. The...
-
Complete the following acid-base reactions: (a) HCCH + NaH
-
Sulfamic acid is a primary standard that can be used to standardize NaOH. Sulfamic acid FM 97.094
-
Cocaine metabolism in rats can be studied by injecting the drug and periodically withdrawing blood to measure levels of metabolites by HPLC-mass spectrometry. For quantitative analysis, isotopically...
-
Explain the difference between systematic and random error.
-
Clausius-Clayperon equation is applicable to (a) Solid-vapour (b) Solid-liquid (c) Liquid-vapour (d) All of these.
-
The reaction \(A(\mathrm{l}) ightarrow R(\mathrm{~g})\) is allowed to reach equilibrium condition in an autoclave. At equilibrium there are two phases - one a pure liquid phase of \(A\) and the other...
-
The equilibrium constant for the reaction \(\mathrm{N}_{2}(\mathrm{~g})+3 \mathrm{H}_{2}(\mathrm{~g}) ightarrow 2 \mathrm{NH}_{3}\) is 0.1084 . Under the same conditions, the equilibrium constant for...

Study smarter with the SolutionInn App


