Treatment of benzene with CO/HCl in the presence of anhydrous AIC1/CuCl followed by reaction with Ac0/NaOAc...
Fantastic news! We've Found the answer you've been seeking!
Question:

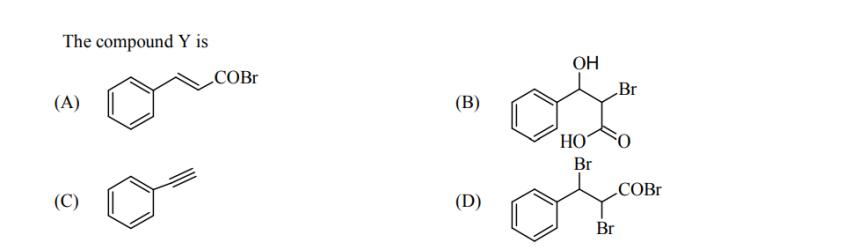
Transcribed Image Text:
Treatment of benzene with CO/HCl in the presence of anhydrous AIC1/CuCl followed by reaction with Ac0/NaOAc gives compound X as the major product. Compound X upon reaction with Br/NaCO3, followed by heating at 473 K with moist KOH furnishes Y as the major product. Reaction of X with H/Pd-C, followed by H3PO4 treatment gives Z as the major product. The compound Y is (A) (C) COBR (B) (D) OH HO Br Br Br O COBR Treatment of benzene with CO/HCl in the presence of anhydrous AIC1/CuCl followed by reaction with Ac0/NaOAc gives compound X as the major product. Compound X upon reaction with Br/NaCO3, followed by heating at 473 K with moist KOH furnishes Y as the major product. Reaction of X with H/Pd-C, followed by H3PO4 treatment gives Z as the major product. The compound Y is (A) (C) COBR (B) (D) OH HO Br Br Br O COBR
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Treatment of tert-butyl alcohol with concentrated HCl gives tert-butyl chloride. When the concentration of is doubled, the reaction rate doubles. When the concentration of tert-butyl alcohol is...
-
Benzene underwent a Friedel-Crafts acylation reaction followed by a Clemmensen reduction. The product gave the following 1H NMR spectrum. What acyl chloride was used in the Friedel-Crafts acylation...
-
Compound A has molecular formula C 8 H 14 O 2 . Upon treatment with catalytic acid, compound. A is converted into the cyclic hemiacetal. Identify the structure of compound A. , Compound A [H*]
-
Diagonalize the matrices, if possible. The eigenvalues are as follows: (11) = 1, 2, 3; (12) = 1, 4; (13) = 5, 1; (14) = 3, 4; (15) = 3, 1; (16) = 2, 1. 3 4 6 1
-
Mack Roe walks to Annas desk where Chip is standing and says, The last program has been tested and incorporated into the system test. The results indicate that the system is finally complete. Every...
-
A polling organization plans to ask a random sample of likely voters who they plan to vote for in an upcoming election. The researchers will report the sample proportion p that favors the incumbent...
-
Consider the patient satisfaction data in Table B.17. For the purposes of this exercise, ignore the regressor "Medical-Surgical." Perform a thorough analysis of these data. Please discuss any...
-
How is it helpful for a private not-for-profit organization to account for current funds as restricted or unrestricted?
-
Please use CX programmer to demonstrate the answer Image transcription text LABWORK 3 PLC 3 - Conveyor & Puncher OBJECTIVES: programing. Design of a WAIT state. Pulse generation function, TOPICS:...
-
Overview The milestone for Project One involves applying accounting principles and methods to long-term liabilities and equity. You will also evaluate these financial statement components for...
-
1. From the observation of one consumer behavior for several months of Year 1. In an economy where the prices of both kinds of goods fluctuate a lot according to the foreign currency exchange rate,...
-
Discuss your responses with your team. What similarities or differences did you find?
-
What is the heat requirement to dilute an inlet aqueous stream of 50% NaOH, by weight, to a fi nal concentration of 10%?
-
Team compensation allows some individuals to work harder than others, yet receive the same pay. Do you agree or disagree? Defend your position.
-
Training programs are frequently the first items eliminated when management wants to cut costs. Why do you believe this occurs?
-
How can an organization develop a competitive advantage internally without the help of outsiders?
-
What is meant by "the real rate of interest"? What is meant by "the nominal rate of interest"? On July 30, 2017, the following information was available to an investor: Yield on 10-year TIPS: 0.58%...
-
A red card is illuminated by red light. What color will the card appear? What if its illuminated by blue light?
-
Glycine has pKa values of 2.3 and 9.6. Would you expect the pKa values of glycylglycine to be higher or lower than these values?
-
Indicate the number of signals and the multiplicity of each signal in the 1H NMR spectrum of each of the following compounds: a. ICH2CH2CH2Br b. ClCH2CH2CH2Cl c. ICH2CH2CHBr2
-
a. Give the product(s) that would be obtained from the reaction of cis-2-butene and trans-2 butene with each of the following reagents. If the products can exist as stereoisomers, show which...
-
Determine the most appropriate strategy that can generate liquidity and accomplish Omos goals. Justify your response. Jing asks Omo about his plans for the shares of his previous employers company....
-
Discuss four important risk and tax-related considerations that are relevant to Omos portfolio. With most of Omos personal net worth tied up in his software company, he is asset rich but cash poor....
-
Recommend, for both Enlows niece and nephew, the most tax-efficient wealth transfer option (lifetime gift or testamentary bequest). Show your calculations. Recommend, for both Enlow's niece and...

Study smarter with the SolutionInn App



