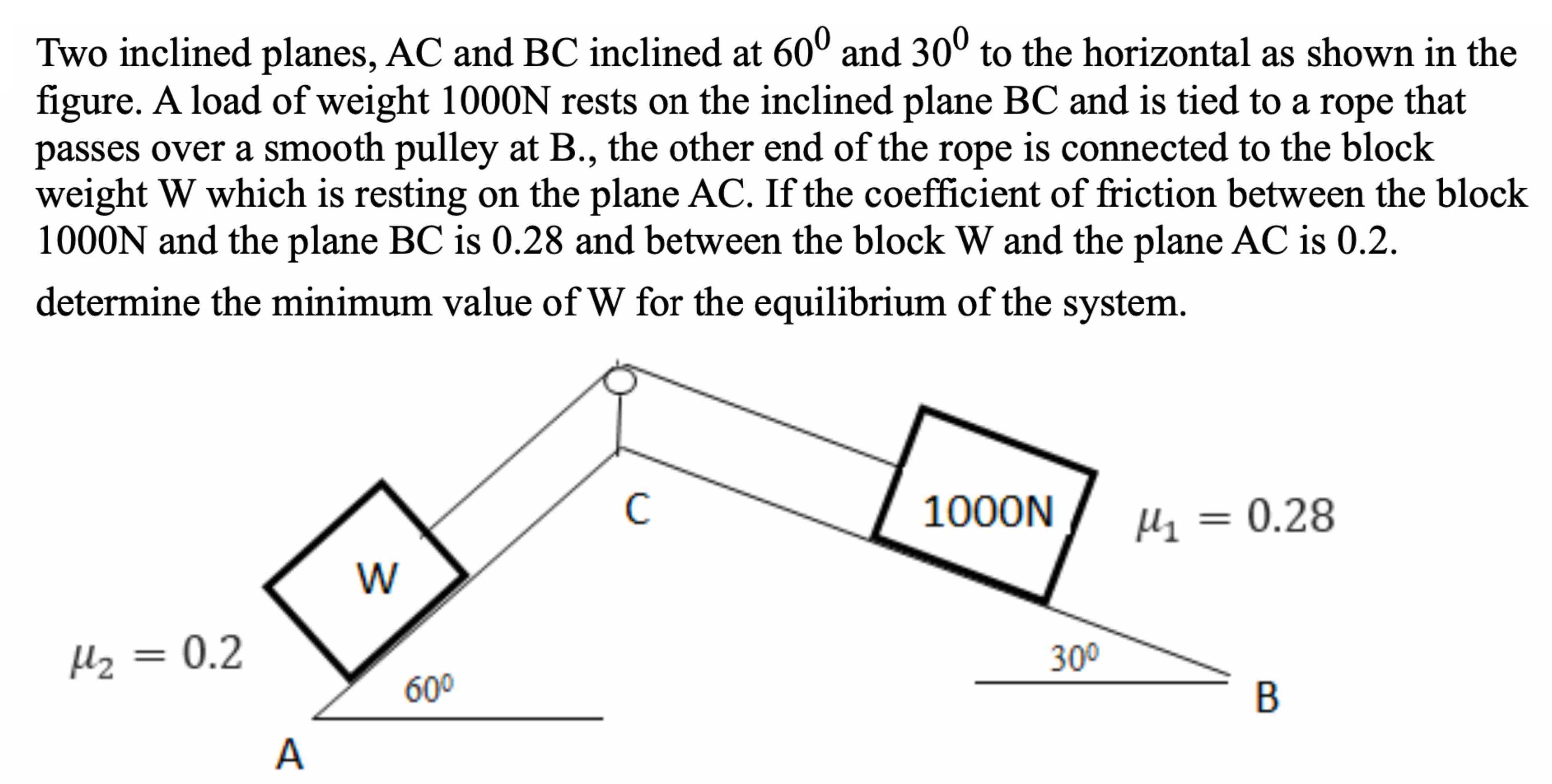
Two inclined planes, AC and BC inclined at 600 and 30 to the horizontal as shown...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Two inclined planes, AC and BC inclined at 600 and 30° to the horizontal as shown in the figure. A load of weight 1000N rests on the inclined plane BC and is tied to a rope that passes over a smooth pulley at B., the other end of the rope is connected to the block weight W which is resting on the plane AC. If the coefficient of friction between the block 1000N and the plane BC is 0.28 and between the block W and the plane AC is 0.2. determine the minimum value of W for the equilibrium of the system. μ₂ = 0.2 A W 60⁰ C 1000N 30⁰ μ₁ = 0.28 B Two inclined planes, AC and BC inclined at 600 and 30° to the horizontal as shown in the figure. A load of weight 1000N rests on the inclined plane BC and is tied to a rope that passes over a smooth pulley at B., the other end of the rope is connected to the block weight W which is resting on the plane AC. If the coefficient of friction between the block 1000N and the plane BC is 0.28 and between the block W and the plane AC is 0.2. determine the minimum value of W for the equilibrium of the system. μ₂ = 0.2 A W 60⁰ C 1000N 30⁰ μ₁ = 0.28 B
Expert Answer:
Answer rating: 100% (QA)
Determine the minimum value of W for the equilibrium of the system We are given that Two inclined planesAC and BCare inclined at 60 and 30 to the hori... View the full answer

Related Book For 

Fundamentals of Ethics for Scientists and Engineers
ISBN: 978-0195134889
1st Edition
Authors: Edmund G. Seebauer, Robert L. Barry
Posted Date:
Students also viewed these corporate finance questions
-
1. Consider a model of revolutions. Society is made up of N > 1 people. Simultaneously, each person chooses whether or not to participate. If n people participate, the probability the revolution...
-
The molar solubility of PbCl in 0.10 M NaCl is 1.7x103 moles per liter (that is, 1.7 x 103 moles of PbCl will dissolve in 1 liter of the solution). What is the Ksp of PbCl? IM Noel + 201 C1 (5) 24
-
Whether the EEOC presented evidence on which a reasonable jury could conclude that Ford retaliated against Harris for filing a charge of discrimination with the EEOC.
-
A companys retained earnings on December 31, 2018 was $2,190,000 and its shareholders equity was $8,760,000. During 2019 the company reported the following: Net income $225,000 A sale of treasury...
-
Describe a conflict situation in which to resolve the problem you would use 1) Avoidance, 2) Defusion, and 3) Confrontation.
-
The file P18_12.txt contains yearly data for the number of licensed drivers (those under 18, those over85, and total) by gender and state. Import this data into Excel and save it as an Excel (.xlsx)...
-
Suppose x is a random variable best described by a uniform probability distribution with c = 20 and d = 45. Find the followmg probabilities: a. ~(20 5 x 5 30) b. ~(20 < x 5 30) c. P(X 2 30) d. ~(x 3...
-
Suppose Intel wishes to raise USD1 billion and is deciding between a domestic dollar bond issue and a Eurobond issue. The U.S. bond can be issued at a 5-year maturity with a coupon of 4.50%, paid...
-
14. What will be the Future worth at the end of five years of a uniform continuous cash flow at the rate P500.00 per year for five years with interest compounded continuously at nominal annual rate...
-
MBA 708 Essentials of Financial Statement Analysis Week 6 Case Study: Transaction and Financial Analysis Tallulah Company has been in business for several years and is publicly traded on a major U.S....
-
Calculate the amount the employer must send to the IRS. Employee's Federal Income Tax $4,848.00 Employee's Social Security $1,945.86 Employee's Medicare $455.08
-
Using a ruler and set squares only, construct the following shapes: a. b. c. d. 5cm 5cm
-
The marketing department has just forecast that 10,000 units of item 778 will be ordered in the next fiscal year. Based on the marketing department's forecast and noting that the seasonal relative...
-
Following are interaction plots for three data sets. Which data set has the largest interactions? Which has the smallest? A B C
-
From your local chamber of commerce, obtain the population figures for your city for the years \(1980,1990,2000\), and 2010. Find the rate of growth for each period. Forecast the population of your...
-
A mass \(m\) is attached at the midpoint of a stretched wire of area of cross-section \(A\), length \(l\), and Young's modulus \(E\) as shown in Fig. 13.29. If the initial tension in the wire is...
-
3: Spring Company is authorized to issue 7500,000 shares of $2 par value common stock. In its first year, the company has the following transactions: March 1: Issued 400,000 shares of stock at $9.75...
-
The nitrogen atoms in N2 participate in multiple bonding, whereas those in hydrazine, N2H4, do not. (a) Draw Lewis structures for both molecules. (b) What is the hybridization of the nitrogen atoms...
-
A particle of rest mass 1 MeV/c 2 and kinetic energy 2 MeV collides with a stationary particle of rest mass 2 MeV/c 2 . After the collision, the particles stick together. Find (a) The speed of the...
-
The relative binding of the extra electron in the arsenic atom that replaces an atom in silicon or germanium can be understood from a calculation of the first Bohr orbit of this electron in these...
-
A pulsed laser fires a 1000-MW pulse of 200-ns duration at a small object of mass 10 mg suspended by a fine fiber 4 cm long. If the radiation is completely absorbed without other effects, what is the...
-
How might the study of organization theory and public management help a public manager who is interested in improving the perception of her agency? LO.1
-
Why do you think citizens of the United States tend to harbor more negative feelings about government compared to citizens of Europe? Support your opinion with research evidence. LO.1
-
How is the systems metaphor useful for understanding organizations? LO.1

Study smarter with the SolutionInn App


