Question: Two small balls with mass 15 g hang in separate thin strings with length L= 1.2 m. The two strings are attached to the
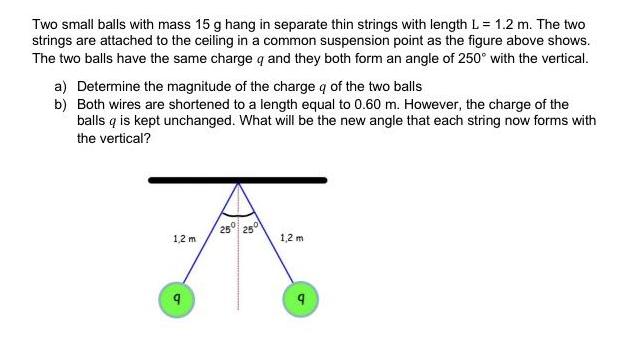
Two small balls with mass 15 g hang in separate thin strings with length L= 1.2 m. The two strings are attached to the ceiling in a common suspension point as the figure above shows. The two balls have the same charge q and they both form an angle of 250 with the vertical. a) Determine the magnitude of the charge q of the two balls b) Both wires are shortened to a length equal to 0.60 m. However, the charge of the balls q is kept unchanged. What will be the new angle that each string now forms with the vertical? 25 25 1,2 m 1,2 m
Step by Step Solution
3.44 Rating (163 Votes )
There are 3 Steps involved in it
A free body diagram is shown below with tension in the strings taken as T I ii The remaining ... View full answer

Get step-by-step solutions from verified subject matter experts


