Draw the organic product(s) you would expect from the following reaction. CH3 CH3 + Br (1...
Fantastic news! We've Found the answer you've been seeking!
Question:
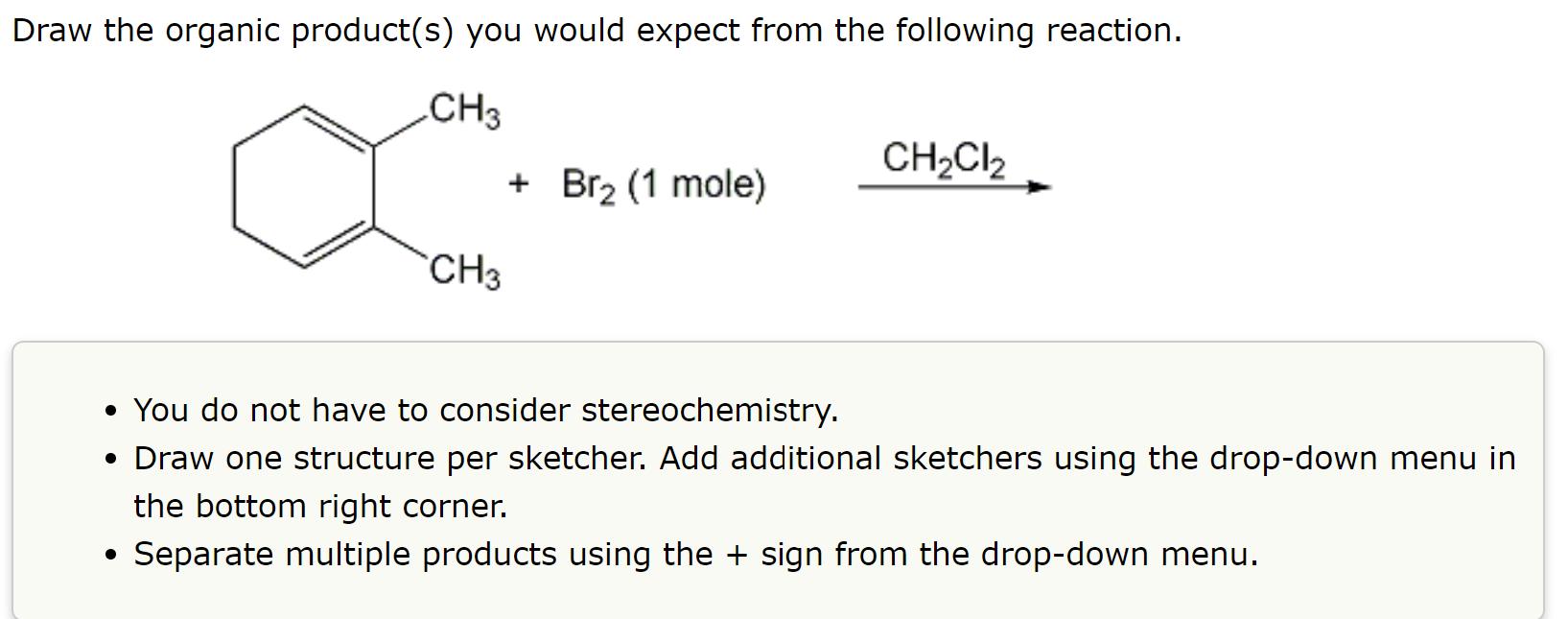
Transcribed Image Text:
Draw the organic product(s) you would expect from the following reaction. CH3 CH3 + Br₂ (1 mole) CH₂Cl2 • You do not have to consider stereochemistry. • Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. Separate multiple products using the + sign from the drop-down menu. Draw the organic product(s) you would expect from the following reaction. CH3 CH3 + Br₂ (1 mole) CH₂Cl2 • You do not have to consider stereochemistry. • Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. Separate multiple products using the + sign from the drop-down menu.
Expert Answer:
Related Book For 

South Western Federal Taxation 2016 Corporations Partnerships Estates and Trusts
ISBN: 9781305399884
39th edition
Authors: James Boyd, William Hoffman, Raabe, David Maloney, Young
Posted Date:
Students also viewed these chemistry questions
-
A company fails to recognize revenue earned but not yet received. Which of the following accounts are involved in the adjusting entry? (a) Asset, (b) Liability, (c) Revenue, or (d) Expense? For the...
-
On December 31, Year 6, Ultra Software Limited purchased 70,000 common shares (70%) of a major competitor, Personal Program Corporation (PPC), at $30 per share. Several shareholders who were...
-
Explain the significance of understanding the internal organisation in context of its general and industry environments.n
-
Suppose a consumer lives in two periods , with his income in period 1 as $100 and his income in period 2 as $150. If the rate of interest in the economy is 12%. Find the equilibrium level of...
-
Use the requirements data from Problem 11 and order costs of $75/order, inventory carrying cost of $1.25/unit/period, and a unit cost of $100 in lots of less than 100 and $95 for lots of 100 or more,...
-
The passage includes all of the following EXCEPT A) Ad hominem B) Verifiable statistics C) Amusing illustration D) Social assessment
-
An electric circuit consists of 7 transistors connected in parallel. Determine the reliability of each transistor if the overall reliability of the circuit is to be 0.99 .
-
Carleton Closures, Inc., manufactures clamps used in the overhead bin latches of several leading airplane models. Greg Poole, president of Carleton Closures, Inc., has gathered the following cost...
-
Mumford Limited had the following financial statement account balances for 2022. Net Sales $356,420 Cost of Goods Sold 152,000 Average Accounts Receivable 19,710 Average Inventory 4,845 average...
-
On January 1, 2017, Hunter Ltd. entered into an agreement to lease a truck from Situ Ltd. Both Hunter and Situ use IFRS 16. The details of the agreement are as follows: Additional information: 1....
-
Let F RM Rh be a linear map with F(x) = Ax and A E M(n x m; R). Let ||xl| the canonical norm of x. Let A with A2:= 71 777 laj 1 i+1 j=1 the so-called canonical Norm on the space M(n x m; R). (You...
-
Match each table with its equation. 1 t Y y = 2 y = x y = VI Y = x Submit Question b. P d. Input -2 Input 2 1 Input -2 0 0 1 2 3 1 2 3 2 3 Input -1 0 1 2 3 Input -2 -1 0 1 2 3 Input -2 -1 0 1 4 9...
-
Write a complete C++ program that asks the user to type two positive integers. Validate the input. It should show a horizontal bar of X symbols whose length is given by the shorter of the two...
-
In the following network, node S transmits packets that pass through a network of 16 routers, and arrive at the destination D. The bit rate of all links is R = 10 Mbit/sec. The packet size is 100...
-
Collaboration in educational institutions usually involves cooperation among students, faculties, sponsors, and universities. Unlike the corporate world, higher education institutions have generally...
-
How innovation is related to technology? Why people interpret innovation differently?
-
I am trying to understand hedging and making a T-Account and I paid for the subscription but I cannot see the explanation of how this works.
-
In each of the following independent cases, document the system using whatever technique(s) your instructor specifies. a. Dreambox Creations (www.dreamboxcreations.com/) in Diamond Bar, California,...
-
Gus (age 84) and Belle (age 18) are married in early 2015. Late in 2015, Belle confronts Gus about his failure to transfer to her the considerable amount of property he previously promised. Gus...
-
Determine whether the source of income for the following sales is U.S. or foreign. a. Suarez, an NRA, sells stock in Home Depot, a U.S. corporation, through a broker in San Antonio. b. Chris sells...
-
Gils outside basis in his interest in the GO Partnership is $100,000. In a proportionate non-liquidating distribution, the partnership distributes to him cash of $30,000, inventory (fair market value...
-
Consider the ammonia process in which \(\mathrm{N}_{2}\) and \(\mathrm{H}_{2}\) (with impurities \(\mathrm{Ar}\) and \(\mathrm{CH}_{4}\) ) are converted to \(\mathrm{NH}_{3}\) at high pressure...
-
Consider the simulation flowsheets in Figure 7.36, which were prepared for ASPEN PLUS. The feed stream, S1, is specified, as are the parameters for each process unit. Complete the simulation...
-
Use a process simulator to determine the flow rate of saturated vapor benzene at \(176.2^{\circ} \mathrm{F}\) and \(1 \mathrm{~atm}\) to be mixed with \(100 \mathrm{lbmol} / \mathrm{hr}\) of liquid...

Study smarter with the SolutionInn App



