The following m/z peaks appeared on a mass spectrum for a peptide. Reconstruct the peptide sequence....
Fantastic news! We've Found the answer you've been seeking!
Question:
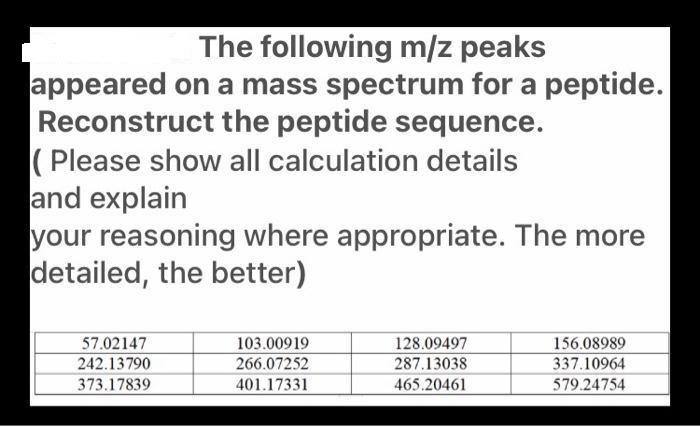
Transcribed Image Text:
The following m/z peaks appeared on a mass spectrum for a peptide. Reconstruct the peptide sequence. (Please show all calculation details and explain your reasoning where appropriate. The more detailed, the better) 57.02147 242.13790 373.17839 103.00919 266.07252 401.17331 128.09497 287.13038 465.20461 156.08989 337.10964 579.24754 The following m/z peaks appeared on a mass spectrum for a peptide. Reconstruct the peptide sequence. (Please show all calculation details and explain your reasoning where appropriate. The more detailed, the better) 57.02147 242.13790 373.17839 103.00919 266.07252 401.17331 128.09497 287.13038 465.20461 156.08989 337.10964 579.24754
Expert Answer:
Answer rating: 100% (QA)
The following peaks we show all calculation details and explam 2 2 A3 Y Y Y3 X... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A concern produces and sells 20,000 units at $100 each. Its variable cost is $50 per unit and fixed cost of concern is $4,00,000. Calculate; (a). P/V ratio, (b). Break-even sales, (c). Break-even...
-
PT ACCESSORY is a company that manufactures mobile accessories, namely: USB data cable, power bank, and stick for taking a photo. The company operates in a monopolistic market. The manager of PT...
-
8. Second Bank has the following balance sheet in millions of dollars, with the Basel risk weights given in parentheses. a. Does the bank have enough capital to meet the requirements as specified by...
-
The time it takes to get a cars oil changed at Speedy Lube is distributed normally with a mean of 12 min and a standard deviation of 2 min. Compute the probability that a customer will have her or...
-
The saving ratio was remarkably stable between 1895 and 1980. When we examine cross-section data, however, we find that the saving ratio tends to rise as income rises. How can these two observations...
-
The common shares of Twitter, Inc. (TWTR) recently traded on the NYSE for $65 per share. You have employee stock options to purchase 1,000 TWTR shares for $70 per share. The options expire in three...
-
The trial balance of Seth Knoll, CPA, P.C., is dated November 30, 2011: During December, Knoll or his business completed the following transactions: Requirements 1. Record the December transactions...
-
1. What are the options for Red Fish-Blue Fish as far as global intermediaries are concerned? What do you recommend? Why? 2. What other options does Red Fish-Blue Fish have to expand their Asian...
-
CoursHeroTranscribedText You have been recruited by a former classmate, Susanna Wu, to join the finance team of a company that she founded recently. The company produces a unique product line of...
-
You and best friend, Doug, are heading out on a 2-week vacation and plan to visit three different destinations. As much as you like Doug, you know that he also has "sticky fingers." He has been...
-
Calculate the Free Cash Flow (FCF) for this company. Income statement, year ended December 31, 2021 (in millions of US dollars, except per share amounts) Sales Net sales Cost of goods sold Gross...
-
Compare the objectives of a product to its features.
-
Compare and contrast components to objects.
-
What is the role of domain analysis in designing a product?
-
Briefly describe how development moves from conceptual modeling to logical and physical designs. Provide one example for a non-IS product and one for an IS product.
-
Describe multiple interfaces and explain why or when they are needed.
-
4. P(2 and N)=- 5. P(less than 4)=6. P(not N)=_ 7. Create a two-way table for the following situation: Brenda's Deli offers a daily brown bag special. The sandwiches choices are ham or turkey and the...
-
In the figure, two loudspeakers, separated by a distance of d1 = 2.63 m, are in phase. Assume the amplitudes of the sound from the speakers are approximately the same at the position of a listener,...
-
Predict the major products of the following reactions, and propose mechanisms to support your predictions. (a) pent-1-ene + HCl (b) 2-methylpropene + HC (c) 1-methylcyclohexene + HI (d)...
-
Predict the products of self-condensation of the following esters. (a) Methyl propanoate + NaOCH3 (b) Ethyl phenylacetate + NaOCH2CH3 (c) (d) CH-C-OCH + NaOCH COOEt +NaOEt
-
Recall (Section 16-10) that two positions of anthracene sometimes react more like polyenes than like aromatic compounds. (a) Draw a Kekulé structure that shows how the reactive positions of...
-
Why are most larger businesses not managed as a single unit by one manager?
-
What are the main advantages and disadvantages that should be considered when deciding between a partnership business and a limited liability company?
-
Suppose an item of information is capable of being provided. It is relevant to a particular decision, it is also reliable, comparable, can be understood by the decision maker concerned and is...

Study smarter with the SolutionInn App


