Use table 17.1 below to calculate G at 355 K for the following equation: 2NaCl(s) + F2(g)
Fantastic news! We've Found the answer you've been seeking!
Question:
Use table 17.1 below to calculate ΔG° at 355 K for the following equation: 2NaCl(s) + F2(g) —> 2NaF(s)+ + Cl2(g)
State whether the equation is spontaneous or not. 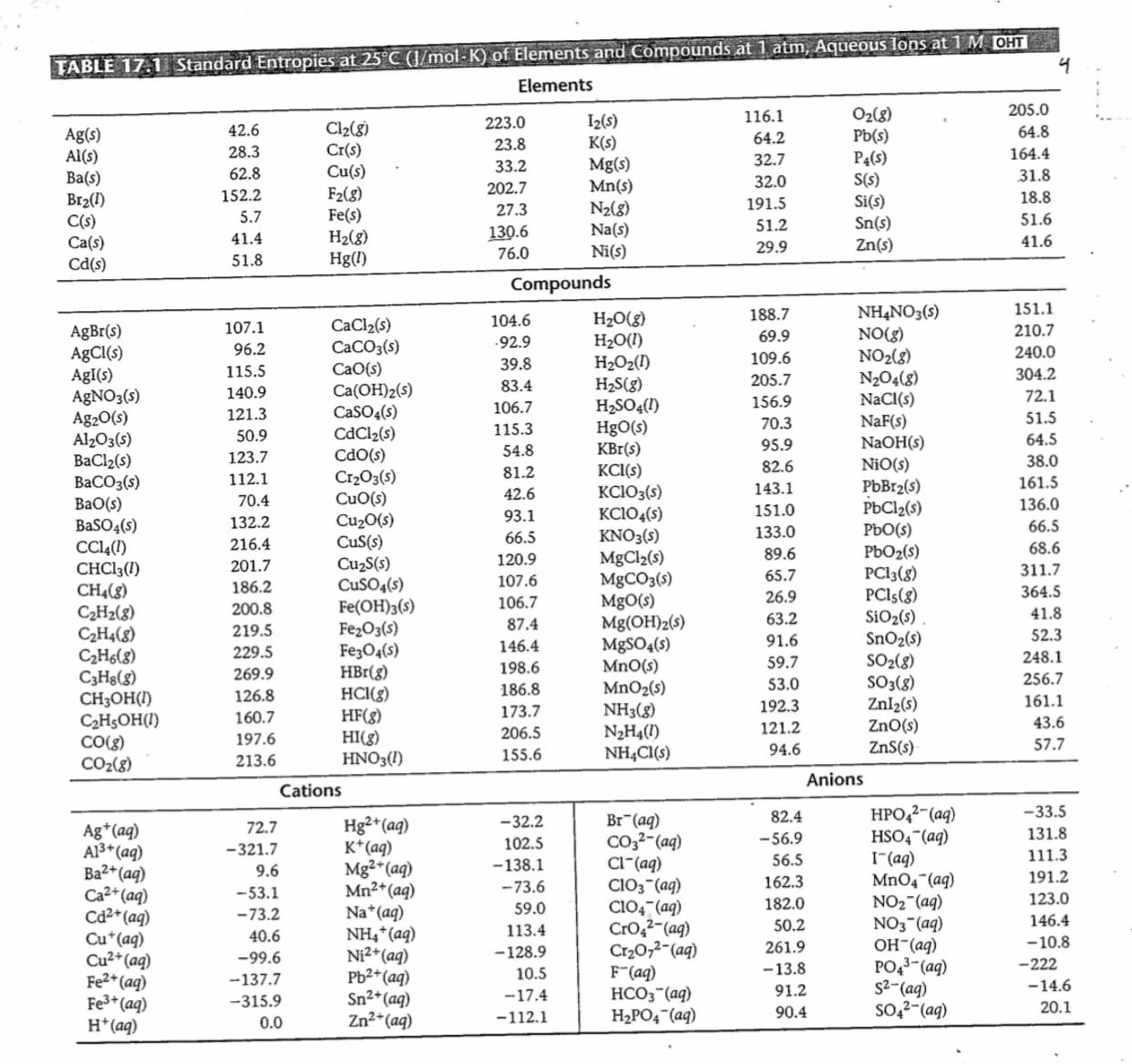
Transcribed Image Text:
TABLE 17.1 Standard Entropies at 25°C (J/mol-K) of Elements and Compounds at 1 atm, Aqueous lons at 1 M OHT Elements 4 205.0 O2(8) Pb(s) P4(s) S(s) Si(s) Sn(s) Zn(s) 116.1 I2(s) K(s) Mg(s) Mn(s) N2(g) Na(s) Ni(s) 223.0 Cl2(8) Cr(s) Cu(s) F2(8) Fe(s) H2(8) Hg(1) 42.6 64.8 Ag(s) Al(s) Ba(s) Br2(1) C(s) Ca(s) Cd(s) 23.8 64.2 28.3 164.4 32.7 32.0 33.2 62.8 31.8 202.7 152.2 18.8 27.3 191.5 5.7 51.6 51.2 130.6 76.0 41.4 41.6 29.9 51.8 Compounds 151.1 NHẠNO3(s) NO(g) NO2(8) N204(8) NaCl(s) 188.7 H20(g) H2O(1) H2O2(1) H2S(g) H2SO4(1) HgO(s) KBr(s) KCI(s) KCIO3(s) KC104(s) KNO3(s) MgCl2(s) MgCO3(s) MgO(s) Mg(OH)2(s) M8SO4(s) MnO(s) MnO2(s) NH3(8) N2H4(1) NH4CI(s) 104.6 CaCl2(s) CaCO3(s) CaO(s) Ca(OH)2(s) CaSO4(s) 107.1 AgBr(s) AgCl(s) AgI(s) AGNO3(s) Ag20(s) Al203(s) BaCl2(s) BaCO3(s) BaO(s) BaSO4(s) CC4(1) CHCI3(1) CH4(8) C2H2(8) C2H4(8) C2H6(8) C3Hs(8) CH3OH(1) C2H5OH(I) CO(8) CO2(8) 69.9 210.7 -92.9 96.2 240.0 39.8 109.6 115.5 304.2 83.4 205.7 140.9 72.1 106.7 156.9 121.3 51.5 115.3 70.3 CdCl2(s) Cdo(s) Cr2O3(s) CuO(s) Cu20(s) CuS(s) CuzS(s) CuSO4(s) Fe(OH)3(s) Fe2O3(s) Fe;O4(s) HBr(g) HCI(g) HF(g) HI(g) HNO3(1) 50.9 64.5 NaOH(s) NiO(s) 54.8 95.9 123.7 112.1 82.6 38.0 81.2 161.5 PbBr2(s) PBC12(s) PbO(s) PbO2(s) PC13(8) PCls(g) SiO2(s) SnO2(s) SO2(8) SO3(8) Znl2(s) ZnO(s) ZnS(s) 42.6 143.1 70.4 136.0 93.1 151.0 132.2 66.5 66.5 133.0 216.4 68.6 120.9 89.6 201.7 311.7 65.7 186.2 107.6 26.9 364.5 106.7 200.8 41.8 87.4 63.2 219.5 52.3 146.4 91.6 229.5 248.1 198.6 59.7 269.9 256.7 186.8 53.0 126.8 161.1 173.7 192.3 160.7 43.6 206.5 121.2 197.6 57.7 155.6 94.6 213.6 Anions Cations HPO,2-(aq) HSO4 (aq) I(aq) MnO4 (aq) NO2 (aq) NO3 (aq) 82.4 -33.5 Hg2*(aq) K*(aq) Mg2* (aq) Mn2+(aq) Na*(aq) NH,*(aq) Ni2*(aq) Pb2+(aq) Sn2*(aq) Zn2*(aq) Br (aq) Co3?-(aq) Cl"(aq) CIO3 (aq) Cl04 (aq) Cro2?-(ag) Cr20,2-(aq) F"(aq) HCO3"(aq) H2PO4 (aq) -32.2 72.7 Ag*(aq) Al3* (aq) Ba2+(aq) Ca2+(aq) Cd2* (aq) Cu*(aq) Cu2* (aq) Fe2* (aq) Fe3+ (aq) H*(aq) -56.9 131.8 -321.7 102.5 56.5 111.3 9.6 -138.1 162.3 191.2 -53.1 -73.6 182.0 123.0 59.0 -73.2 146.4 113.4 50.2 40.6 (bv)_HO PO43-(ag) 261.9 -10.8 -99.6 -128.9 10.5 -13.8 -222 -137.7 (bv)-2S (bv)-z*os 91.2 -14.6 -17.4 -315.9 90.4 20.1 0.0 -112.1 TABLE 17.1 Standard Entropies at 25°C (J/mol-K) of Elements and Compounds at 1 atm, Aqueous lons at 1 M OHT Elements 4 205.0 O2(8) Pb(s) P4(s) S(s) Si(s) Sn(s) Zn(s) 116.1 I2(s) K(s) Mg(s) Mn(s) N2(g) Na(s) Ni(s) 223.0 Cl2(8) Cr(s) Cu(s) F2(8) Fe(s) H2(8) Hg(1) 42.6 64.8 Ag(s) Al(s) Ba(s) Br2(1) C(s) Ca(s) Cd(s) 23.8 64.2 28.3 164.4 32.7 32.0 33.2 62.8 31.8 202.7 152.2 18.8 27.3 191.5 5.7 51.6 51.2 130.6 76.0 41.4 41.6 29.9 51.8 Compounds 151.1 NHẠNO3(s) NO(g) NO2(8) N204(8) NaCl(s) 188.7 H20(g) H2O(1) H2O2(1) H2S(g) H2SO4(1) HgO(s) KBr(s) KCI(s) KCIO3(s) KC104(s) KNO3(s) MgCl2(s) MgCO3(s) MgO(s) Mg(OH)2(s) M8SO4(s) MnO(s) MnO2(s) NH3(8) N2H4(1) NH4CI(s) 104.6 CaCl2(s) CaCO3(s) CaO(s) Ca(OH)2(s) CaSO4(s) 107.1 AgBr(s) AgCl(s) AgI(s) AGNO3(s) Ag20(s) Al203(s) BaCl2(s) BaCO3(s) BaO(s) BaSO4(s) CC4(1) CHCI3(1) CH4(8) C2H2(8) C2H4(8) C2H6(8) C3Hs(8) CH3OH(1) C2H5OH(I) CO(8) CO2(8) 69.9 210.7 -92.9 96.2 240.0 39.8 109.6 115.5 304.2 83.4 205.7 140.9 72.1 106.7 156.9 121.3 51.5 115.3 70.3 CdCl2(s) Cdo(s) Cr2O3(s) CuO(s) Cu20(s) CuS(s) CuzS(s) CuSO4(s) Fe(OH)3(s) Fe2O3(s) Fe;O4(s) HBr(g) HCI(g) HF(g) HI(g) HNO3(1) 50.9 64.5 NaOH(s) NiO(s) 54.8 95.9 123.7 112.1 82.6 38.0 81.2 161.5 PbBr2(s) PBC12(s) PbO(s) PbO2(s) PC13(8) PCls(g) SiO2(s) SnO2(s) SO2(8) SO3(8) Znl2(s) ZnO(s) ZnS(s) 42.6 143.1 70.4 136.0 93.1 151.0 132.2 66.5 66.5 133.0 216.4 68.6 120.9 89.6 201.7 311.7 65.7 186.2 107.6 26.9 364.5 106.7 200.8 41.8 87.4 63.2 219.5 52.3 146.4 91.6 229.5 248.1 198.6 59.7 269.9 256.7 186.8 53.0 126.8 161.1 173.7 192.3 160.7 43.6 206.5 121.2 197.6 57.7 155.6 94.6 213.6 Anions Cations HPO,2-(aq) HSO4 (aq) I(aq) MnO4 (aq) NO2 (aq) NO3 (aq) 82.4 -33.5 Hg2*(aq) K*(aq) Mg2* (aq) Mn2+(aq) Na*(aq) NH,*(aq) Ni2*(aq) Pb2+(aq) Sn2*(aq) Zn2*(aq) Br (aq) Co3?-(aq) Cl"(aq) CIO3 (aq) Cl04 (aq) Cro2?-(ag) Cr20,2-(aq) F"(aq) HCO3"(aq) H2PO4 (aq) -32.2 72.7 Ag*(aq) Al3* (aq) Ba2+(aq) Ca2+(aq) Cd2* (aq) Cu*(aq) Cu2* (aq) Fe2* (aq) Fe3+ (aq) H*(aq) -56.9 131.8 -321.7 102.5 56.5 111.3 9.6 -138.1 162.3 191.2 -53.1 -73.6 182.0 123.0 59.0 -73.2 146.4 113.4 50.2 40.6 (bv)_HO PO43-(ag) 261.9 -10.8 -99.6 -128.9 10.5 -13.8 -222 -137.7 (bv)-2S (bv)-z*os 91.2 -14.6 -17.4 -315.9 90.4 20.1 0.0 -112.1
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Calculate E, G , and K for the following reactions. (a) 4Co3+ + 2H2O 4Co2+ O2(g) + 4H+ (b) Ag(S2O3)32- + Fe(CN)4-6 Ag(s) + 2S2O2- 3 + Fe(CN)3-6
-
Calculate H o R and U o R at 298.15 K for the following reactions: a. 4NH 3 (g) + 6NO(g) 5N 2 (g) + 6H 2 O(g) b. 2NO(g) + O 2 (g) 2NO 2 (g) c. TiCl 4 (l) + 2H 2 O(l) TiO 2 (s) + 4HCl(g) d....
-
Calculate the equilibrium constant K for the following reaction at 25C from standard electrode potentials. Fe3 + (aq) + Cu(s) Fe2+(aq) + Cu2+(aq) The equation is not balanced.
-
In Problems 530, a. Classify the sequences as arithmetic, geometric, Fibonacci, or none of these. b. If arithmetic, give d; if geometric, give r; if Fibonacci, give the first two terms; and if none...
-
Explain the differences between buy-side and sell-side e-procurement. Give an advantage for each type for the purchasing company.
-
Ms. Espinoza is retired and depends on her investments for her income. Mr. Liu is a young executive who wants to save for the future. Both are stockholders in Scaled Composites, LLC, which is...
-
The type of model building used by Pauling and by Watson and Crick involved the use of ball-and-stick units. Model building can now be done with computer software. Even though you may not be familiar...
-
True or False. Write T if the corresponding statement is true. If the statement is false, write F and state what changes should be made to make it a true statement. 1. Activities of a general purpose...
-
dentify the correctly revised sentence that adds interest and variety by beginning with an infinitive. Brooke congratulated me on securing investor financing for my startup company. Multiple choice...
-
A piezoelectric force sensor has the following configuration (shown in Fig.02). If a force is applied in the direction shown, what will be the expression of the output voltage? 3A 'piezo (a) Lpiezo...
-
Over 350 students took a college calculus final exam. The scores of the students follow a normal distribution. Using the information given below, determine the mean and the standard deviation for the...
-
Gaterburg Hospitals general ledger contains the following unadjusted account balances, among others, for December 31, 20X5: Acct. No. 104 Accounts receivable $120,000 105 Allowance for uncollectible...
-
An American exporter denominates its Australian exports in AUD and expects to receive AUD 2,000,000 in 1 year. The company's financial manager expects the AUD rate after 1 year to be 1.40. The Firm...
-
Select one of your favorite retailers. Research the following topics in detail with thoroughness and completion, and include into a typed minimum 4 page report. In addition, you will need to include...
-
You are given the following information: EUR deposit rate for 1 year 2% EUR borrowing rate for 1 year Australian Dollar deposit rate for 1-year Australian Dollar borrowing rate for 1 year AUD/EUR...
-
On January 1, 2025, Wildhorse Corporation had the following stockholders' equity accounts. Common Stock ($10 par value, 50,000 shares issued and outstanding) $500,000 Paid-in Capital in Excess of...
-
Create a star schema for analyzing sales data using PostgreSQL. The data model will contain: Product dimension: it contains information about each product, product ID, product name, number,...
-
7. FALSE DILEMMA 8. GANDWAGON Definition: Fallacy example: How to revise argument: Definition: Fallacy example: How to revise argument:
-
Titanocene dichloride, (-C5H5)2TiCl2, is a potential antitumor drug thought to be carried to cancer cells by the protein transferring (Figure 17-7). To measure the Ti(IV) binding capacity of...
-
(a) Use Equations 8-20 and 8-21 to find the pH and concentrations of HA and A- in a solution prepared by mixing 0.00200 mol of acetic acid plus 0.004 00 mol of sodium acetate in 1.00 L of water. (b)...
-
The weak base B (Kb = 1.0 10-5) equilibrates between water (phase 1) and benzene (phase 2). (a) Define the distribution coefficient, D, for this system. (b) Explain the difference between D and K,...
-
Figure 1.10 shows an automatic water level control system. (a) Explain how the automatic control system works indicating the roles of key components. (b) If the automatic water level control system...
-
Many closed-loop and open-loop control systems can be found in homes. (a) List six such examples (three open-loop and three closed-loop). (b) Construct feedback control system block diagrams for the...
-
The diagram depicted in Figure 1.38 is a closed-loop temperature control system. (a) Explain how this control system works. (b) If the automatic controller is replaced by manual control, explain how...

Study smarter with the SolutionInn App


