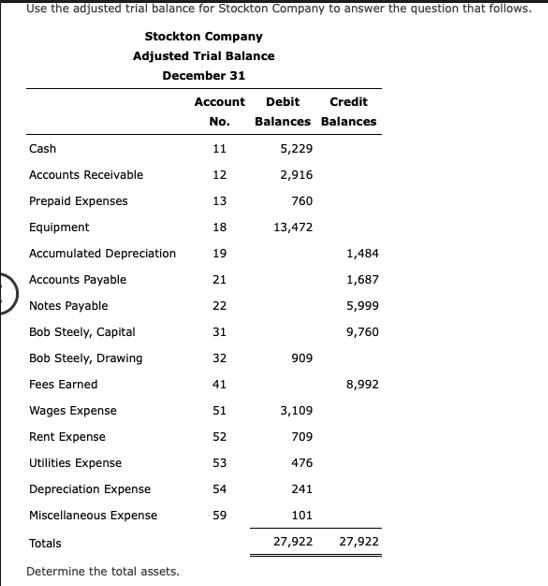
Use the adjusted trial balance for Stockton Company to answer the question that follows. Stockton Company...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Use the adjusted trial balance for Stockton Company to answer the question that follows. Stockton Company Adjusted Trial Balance December 31 Cash Accounts Receivable Prepaid Expenses Equipment Accumulated Depreciation Accounts Payable Notes Payable Bob Steely, Capital Bob Steely, Drawing Fees Earned Wages Expense Rent Expense Utilities Expense Depreciation Expense Miscellaneous Expense Totals Determine the total assets. Account Debit Credit No. Balances Balances 11 12 13 18 19 21 22 31 32 41 51 52 53 54 59 5,229 2,916 760 13,472 909 3,109 709 476 241 101 27,922 1,484 1,687 5,999 9,760 8,992 27,922 Use the adjusted trial balance for Stockton Company to answer the question that follows. Stockton Company Adjusted Trial Balance December 31 Cash Accounts Receivable Prepaid Expenses Equipment Accumulated Depreciation Accounts Payable Notes Payable Bob Steely, Capital Bob Steely, Drawing Fees Earned Wages Expense Rent Expense Utilities Expense Depreciation Expense Miscellaneous Expense Totals Determine the total assets. Account Debit Credit No. Balances Balances 11 12 13 18 19 21 22 31 32 41 51 52 53 54 59 5,229 2,916 760 13,472 909 3,109 709 476 241 101 27,922 1,484 1,687 5,999 9,760 8,992 27,922
Expert Answer:
Answer rating: 100% (QA)
To determine the total assets for Stockton Company you ne... View the full answer

Related Book For 

Interpreting and Analyzing Financial Statements
ISBN: 978-0132746243
6th edition
Authors: Karen P. Schoenebeck, Mark P. Holtzman
Posted Date:
Students also viewed these accounting questions
-
Supporters of globalization claim it has a positive impact on long-run economic growth. Opponents may point to its costs. Evaluate the impact of globalization, using Apple or any other organization...
-
Analyze the given database tables I. ANALYSIS. Analyze the given cases and answer the questions 1. Given the Un-normalized Student table, analyze by steps the process of converting this table to 1NF...
-
Everest plc is considering investing in a number of new investment projects - A, B, and C. The expected pattern of net cash flows for each project are: B 0 1 (800) (950) 200 250 200 250 200 290 350...
-
EVE, Inc. reported a return on investment of 20%, a sales margin of 25%, and residual income of $480,000. The required rate of return on invested capital is 8%. On the basis of this information, the...
-
The van't Hoff equation relates the chemical equilibrium constant K to the enthalpy of reaction Ho. From the value of K in Table A.10 for the dissociation of hydrogen at 2000 K and the value...
-
In the figure for Prob. 161, the inside rim diameter is 280 mm and the dimension R is 90 mm. The shoes have a face width of 30 mm. Find the braking torque and the maximum pressure for each shoe if...
-
Internal auditors focuson-;external auditors are more concerned with -.(Fill in the blanks.) a. financial statements; risk assesment b. company policies and procedures; financial statements C....
-
Canyon Canoe Company has experienced rapid growth in its first few months of operations and has had a significant increase in customers renting canoes and purchasing T-shirts. Many of these customers...
-
Should a retail store management provide different layouts for different genders? Discuss.
-
5. Suppose an investor invests in a portfolio with price S and constant dividend yield q. Assume the investor is charged a constant expense ratio a (which acts as a negative dividend) and at date T...
-
In this assessment, you will work in a group to critically analyse beliefs about children and early childhood practice and creatively reimagine how this might look in practice. This will be in the...
-
Charlotte - Mecklenburg County's Health Department has hired your team to do a community assessment of root causes of a recent rash of high school student drug overdoses toward the goal of creating...
-
Josie Ferreira opened a medical practice. During July, the first month of operation, the business, titled J. Ferreira, MD, experienced the following events: View the transactions. Analyze the effects...
-
In the equation, QPR is operating revenues for firm in year t, defined as the sum of revenues from sales and services and other revenues generated from operating activities; TAL-I is total lagged...
-
1)Q)What is Servlet ? 2)What is web application? 3)What is web resource program? 4) What is Web container? 5) What is ServletConfig object? 6)What is ServletContext object? 7) what is Servlet...
-
Use the link provided to read the article entitled "Childhood Denied" to gain insight into the human rights violations faced by indigenous peoples of Canada. Additionally, watch the videos included...
-
Decades after the event, Johnson & Johnson (J&J), the 130-year-old American multinational, is still praised for swiftly The company indicated that its response was based on the expectations set forth...
-
Q1 McDonalds is the worlds (________ / #2) restaurant chain when measured by (________/ # of units) and has more than 32,000 restaurants in more than 120 countries. Q2. In regard to assets, this...
-
Q1. Sales Revenue minus (operating expenses / non-operating revenues and expenses / ____________equals Gross Profit that totals ____________million. Q2. Gross profit minus (provision for income tax /...
-
Q1. Wal-Mart generates profits from a (high mark up / _______________). What other companies have a low-cost high-volume strategy? (__________ / __________ /DELL / AAPL) Contributing to CC and BBYs...
-
In an ideal gas mixture consisting of components \(A\) and \(B\), the mole fraction of component \(A\) is equal to (a) \(n_{\mathrm{A}}=\frac{p_{\mathrm{A}}+p_{\mathrm{B}}}{P}\) (b)...
-
The availability of a system (a) Depends upon the conditions of the system only (b) Is independent of the conditions of the surroundings (c) Does not depend upon the conditions of the system (d)...
-
In an absorption refrigeration system, the heat energy \(Q_{1}\) is supplied at temperature \(T_{1}\) while the system absorbs heat energy \(Q_{3}\) from a cold space at temperature \(T_{3}\). If the...

Study smarter with the SolutionInn App


