Use the Fe-C phase diagram at the end to answer the following questions. A 0.55 wt%...
Fantastic news! We've Found the answer you've been seeking!
Question:


Transcribed Image Text:
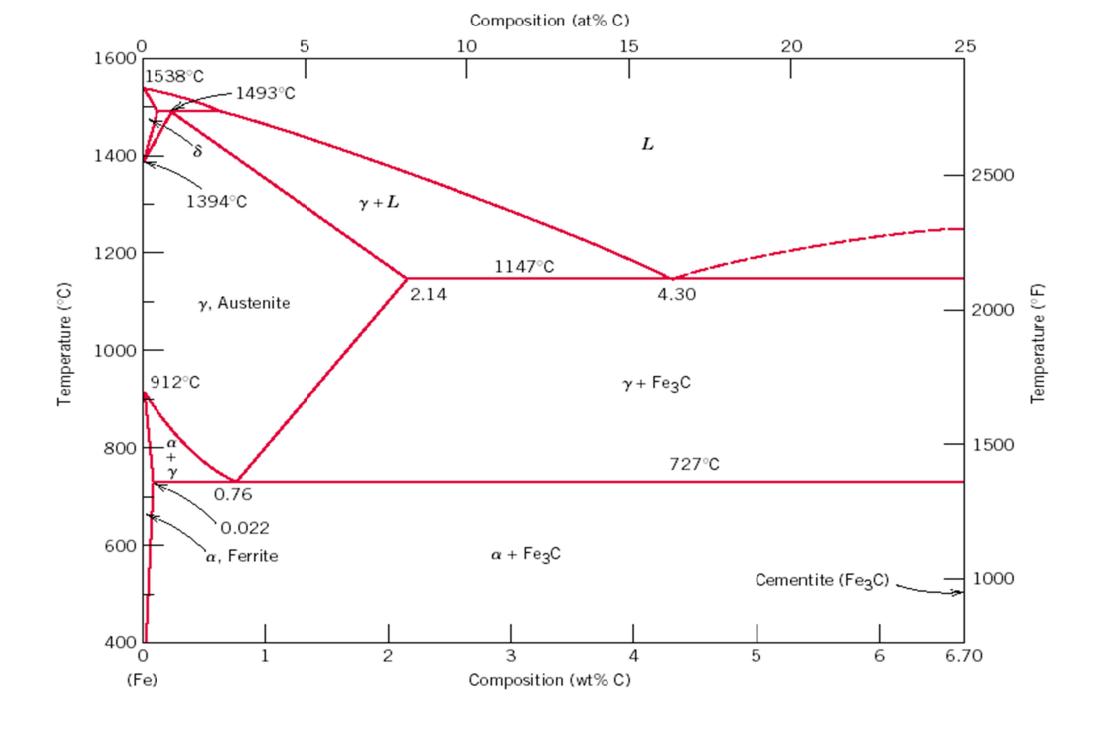
Use the Fe-C phase diagram at the end to answer the following questions. A 0.55 wt% carbon steel is slow cooled from 1000°C to a temperature slightly below 727°C. In the final structure: (a) Sketch and label a typical microstructure showing all phases & micro-constituents that would be present at 720 °C (b) Indicate the phase or phases present and the composition of each phase, and calculate how much of each phase is present. (c) At the same temperature determine the fractions of (proeutectoid) ferrite and pearlite. (d) What phase would be present if the sample is very rapidly quenched instead from 1000°C to room temperature? (e) Can this phase be found on the Fe-C diagram, and if not, why not? Temperature (°C) 0 1600 1400 1200 1000 1538°C 600 800 a 400 0 912°C (Fe) +2 8 Y 1493°C 1394°C y, Austenite 0.76 0.022 a, Ferrite 5 y + L 2 2.14 Composition (at% C) 15 10 1147°C a + Fe3C L 3 4 Composition (wt% C) 4.30 y + Fe3C 727°C 20 Cementite (Fe3C) 5 6 25 2500 2000 1500 1000 6.70 Temperature (°F) 1. Use the Fe-C phase diagram above to answer the following questions. A 0.55 wt% carbon steel is slow cooled from 1000°C to a temperature slightly below 727°C. In the final structure: a. Sketch and label a typical microstructure showing all phases & micro-constituents that would be present at 720 °C. b. Indicate the phase or phases present and the composition of each phase, and calculate how much of each phase is present. c. At the same temperature determine the fractions of (proeutectoid) ferrite and pearlite. d. What phase would be present if the sample is very rapidly quenchedinstead from 1000°C to room temperature? e. Can this phase be found on the Fe-C diagram, and if not, why not? Use the Fe-C phase diagram at the end to answer the following questions. A 0.55 wt% carbon steel is slow cooled from 1000°C to a temperature slightly below 727°C. In the final structure: (a) Sketch and label a typical microstructure showing all phases & micro-constituents that would be present at 720 °C (b) Indicate the phase or phases present and the composition of each phase, and calculate how much of each phase is present. (c) At the same temperature determine the fractions of (proeutectoid) ferrite and pearlite. (d) What phase would be present if the sample is very rapidly quenched instead from 1000°C to room temperature? (e) Can this phase be found on the Fe-C diagram, and if not, why not? Temperature (°C) 0 1600 1400 1200 1000 1538°C 600 800 a 400 0 912°C (Fe) +2 8 Y 1493°C 1394°C y, Austenite 0.76 0.022 a, Ferrite 5 y + L 2 2.14 Composition (at% C) 15 10 1147°C a + Fe3C L 3 4 Composition (wt% C) 4.30 y + Fe3C 727°C 20 Cementite (Fe3C) 5 6 25 2500 2000 1500 1000 6.70 Temperature (°F) 1. Use the Fe-C phase diagram above to answer the following questions. A 0.55 wt% carbon steel is slow cooled from 1000°C to a temperature slightly below 727°C. In the final structure: a. Sketch and label a typical microstructure showing all phases & micro-constituents that would be present at 720 °C. b. Indicate the phase or phases present and the composition of each phase, and calculate how much of each phase is present. c. At the same temperature determine the fractions of (proeutectoid) ferrite and pearlite. d. What phase would be present if the sample is very rapidly quenchedinstead from 1000°C to room temperature? e. Can this phase be found on the Fe-C diagram, and if not, why not?
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Paula makes the following acquisitions of ordinary shares in Indigent plc: She sells 500 shares on 28 March 2021. No shares are acquired within the next 30 days. (a) Show the s104 holding on 28 March...
-
Do producers tend to favor price floors or price ceilings? Briefly explain.
-
Consider a single-server queueing system with a finite queue that can hold a maximum of 2 customers excluding any being served. The server can provide batch service to 2 customers simultaneously,...
-
Custom Baseball Hats sells all its merchandise on credit. It has a profit margin of 4 percent, days sales outstanding equal to 60 days (based on a 365-day year), receivables of $\$ 147,945.20$, total...
-
Lucile Bluth has been asked by the Board of Directors to provide a brief presentation at the upcoming meeting to identify the root cause of project G.O.B's challenges as well as identify a feasible...
-
SHA E SI SINE THIS IS A TEST ASYL "NASA completed a record-breaking experiment last week, and it involved streaming an adorable cat video from 19 million miles away (about 80 times the distance...
-
Paoli Consulting is a consulting firm owned and operated by Mary Paoli. The following end-of-period spreadsheet was prepared for the year ended March 31, 2019: During the year ended March 31, 2019,...
-
Create a python program that evaluates the following equations in a created file : A = B Output Sample: (0.0, 5.2) lies on the y-axis (4.0, 0.0) lies on the x-axis (-2.0, 5.2) is on Quadrant II...
-
A cube of mass m, glides through from rest along a frictionless quarter ramp from height 6 m and then crashes with stationary cube of mass m (m-0.5m). After the impact (elastic collision), the second...
-
3. An object m is tied to one end of a string, moves in a circle with a constant speed on a horizontal frictionless table. The second end of the string is connected to a big mass M and goes through a...
-
(b) State Carnot's theorem. Draw the PV diagram showing the Carnot's cycle for an ideal gas and arrive at IQH/Qcl = TH/Tc (3 M)
-
a) Construct a graph of Weight versus Spring Displacement with Spring Displacement on the x-axis. Draw the line that best fits your data points. The slope of the graph is the spring constant for the...
-
Consider a classical gas consisting of N impenetrable hard sphere particles of radius R. If the gas is placed in a relatively large container of volume V, a. (5%) determine the dependence of (N,V, E)...
-
Mrs. Hop is going to open a music store with CDs. She has got CZK 180 000 from her savings, which she invests into her business and she has also received a bank loan in the amount of CZK 70 000. She...
-
Chloroplasts are illuminated until the levels of the Calvin cycle intermediates reach a steady state. The light is then turned off. How does the level of RuBP vary after this point?
-
The following elementary reactions are to be carried out in a PFR with a heat exchange with constant Ta: 2A+BCHRx1B=10kJmol BADHRx2A=+10kJmol AB+2CEHRx3C=20kJmol C The reactants all enter at 400 K....
-
Use Equation (3-20) to make a plot of f(E,T) as a function of E for T = 300, 500, 800, and 1200 K. a. What is the fraction of molecules that have sufficient energy to pass over a energy barrier of 25...
-
The elementary reversible reaction 2A B is carried out in a flow reactor where pure A is fed at a concentration of 4.0 mol/dm 3 . If the equilibrium conversion is found to be 60%, a. What is the...
-
Explain the significance of \(g_{c}\) in Newton's second law. What are the magnitude and units of \(g_{c}\) in the English Engineering system? In the SI system?
-
Explain the difference between absolute and gage pressures.
-
Nitrogen gas is reversibly compressed from \(70^{\circ} \mathrm{F}\) and 14.7 psia to one-fourth of its original volume by (1) a \(T=\) const process or (2) a \(p=\) const process followed by a...

Study smarter with the SolutionInn App


