use the ff table for answering Tank 1 containing pure carbon monoxide (A) at 2 atm is
Fantastic news! We've Found the answer you've been seeking!
Question:

use the ff table for answering


Transcribed Image Text:
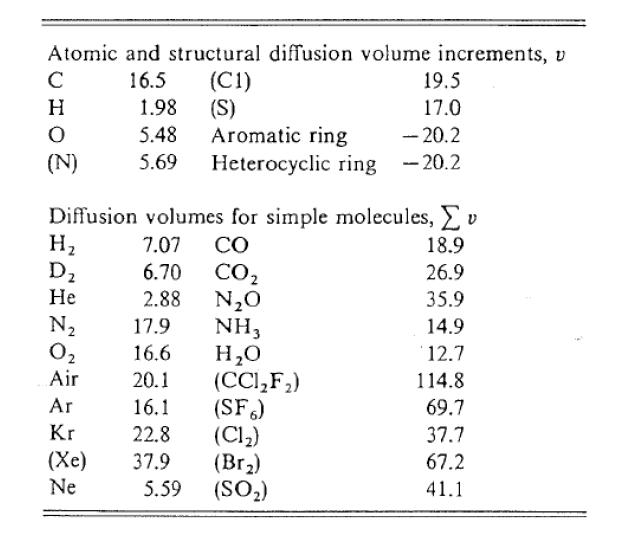
Tank 1 containing pure carbon monoxide (A) at 2 atm is connected to another tank, Tank 2, containing pure hydrogen gas (B) also at 72 kPa. Both tanks are at 15 o C. Initially, the valve located at the 0.72132- in diameter, 200-mm long tube connecting the two tanks is closed to prevent mixing. Both tanks have a volume of 50 cubic feet. However, due to mistakes by the ones monitoring the tanks, the valve was inadvertently opened and mixing of the gases commences. At the time they noticed that the valve was open, the mass fraction of CO in one tank is 0.49987 and the valve was immediately closed. Assume ideal gas behavior. (Atomic weights: C-12.00, H-1.00, 0-16.00) Dco-H₂ Determine using the appropriate correlation. What is the mole fraction of H2 in Tank 1 after the valve was closed, assuming the tube volume is negligible? What is the total molar rate of diffusion of H2 through the tube right before the valve was closed in mol/s? Towards which tank is H2 diffusing? Tank 1 containing pure carbon monoxide (A) at 2 atm is connected to another tank, Tank 2, containing pure hydrogen gas (B) also at 72 kPa. Both tanks are at 15 o C. Initially, the valve located at the 0.72132- in diameter, 200-mm long tube connecting the two tanks is closed to prevent mixing. Both tanks have a volume of 50 cubic feet. However, due to mistakes by the ones monitoring the tanks, the valve was inadvertently opened and mixing of the gases commences. At the time they noticed that the valve was open, the mass fraction of CO in one tank is 0.49987 and the valve was immediately closed. Assume ideal gas behavior. (Atomic weights: C-12.00, H-1.00, 0-16.00) Dco-H₂ Determine using the appropriate correlation. What is the mole fraction of H2 in Tank 1 after the valve was closed, assuming the tube volume is negligible? What is the total molar rate of diffusion of H2 through the tube right before the valve was closed in mol/s? Towards which tank is H2 diffusing?
Expert Answer:
Posted Date:
Students also viewed these chemical engineering questions
-
use the ff table to answer A 0.72132-cm diameter spherical ball of para-dichlorobenzene (p-DCB, C6H4C12), the main component of deodorant in restrooms, is left suspended in air in a large room at 20...
-
Use Table FA-1 (in Exhibit B-2) and Table FA-2 (in Exhibit B-4) to determine the future amounts of the following investments. Table FA-1 Table FA-2 a. $60,000 is invested for 10 years, at 6 percent...
-
Use bond energy values in Table to estimate ÎH for each of the following reactions in the gas phase. a. H2(g) + Cl2(g) 2HCl(g) Table s | 14 39 95 45 72 1 1419 6847064968 77386 42222 34 985 0302...
-
Copper Industries (a sole proprietorship) sold three 1231 assets during 2015. Data on these property dispositions are as follows: a. Determine the amount and the character of the recognized gain or...
-
The table shows 2008 GSS data on family income and the subjects highest degree attained, for which X2 = 315.98 (P-value = 0.000). a. Estimate the difference between the lowest and highest education...
-
What is the difference between the "retained earnings" number on the income statement and the balance sheet?
-
A long straight wire carries a current of magnitude \(I\), and this current creates a magnetic field \(\vec{B}\). Derive an expression for the magnitude of the magnetic field a radial distance \(r\)...
-
The following accounts and balances were drawn from the records of Barker Company at December 31, 2016: Supplies ...............$ 1,000 Beginning retained earnings .......$ 9,300 Cash flow from...
-
6. The major components in "Gun Metal" are: (a) Cu, Ni and Fe (b) Al, Cu, Mg and Mn (c) Cu, Sn and Zn 7. 8. Which of the following ore is concentrated using group 1 cyanide salt (a) Calamine (b)...
-
John Wallace is an automotive enthusiast. He has over 25 years of experience as a mechanic for the dealership of a large car manufacturer in Oakville. John also gained experience doing minor body...
-
Apache's real estate department is considering buying a hangar and leasing it out to private jet operators. They ask you to calculate the NPV and IRR of the investment and have given you the data...
-
11.12 Water, oil, and an unknown fluid are contained in the open vertical tubes shown in Fig. P11.12. Determine the den- sity of the unknown fluid. 1-in. diameter Oil, SG = 0.9 1 ft 1 I Water Figure...
-
A parallel circuit has a load made up from a resistance of 4092 and an inductive reactance of 2592, and is connected to an a.c. 50Hz supply. Calculate the voltage required to give an output power of...
-
0.3 m 12 rad/s 0.6 m C. 30 Based on the above figure, locate the location of instantaneous Center (IC) of zero velocity. a. Point A b. Point B Point O O d. None of the mentioned Clear my choice OB
-
B. The flow of a river of 21.25 m/sec produces a total brake power of 5400 KW. It is proposed to install two turbines, one of which is twice the capacity of the other. The efficiency and specific...
-
8.2 Fixed temperature points in the International Temperature Scale are phase equilibrium states for a variety of pure substances. Discuss the conditions necessary within an experimental apparatus to...
-
The Constantine Corporation purchases robes for $12 per unit and then sells them for $25 per unit. The company estimated the following sales for the first four months of the current year: January...
-
At Glass Company, materials are added at the beginning of the process and conversion costs are added uniformly. Work in process, beginning: Number of units Transferred - in costs Direct materials...
-
Grill Master Company sells total outdoor grilling solutions, providing gas and charcoal grills, accessories, and installation services for custom patio grilling stations. Instructions Respond to the...
-
Refer to the revenue arrangement in P18-1. Instructions Consider the following information and respond to the requirements indicated. (a) In response to competitive pressures for the Internet access...
-
Tablet Tailors sells tablet PCs combined with Internet service (Tablet Bundle A) that permits the tablet to connect to the Internet anywhere (set up a Wi-Fi hot spot). The price for the tablet and a...

Study smarter with the SolutionInn App


