Question: Use the pH lab to sort each item into the proper bin. 0.1 M HCI 0.1 M HCH302 0.1 M NaOH 0.1 M NH3
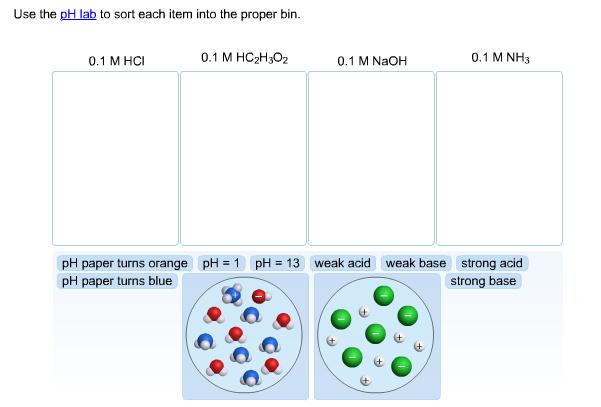
Use the pH lab to sort each item into the proper bin. 0.1 M HCI 0.1 M HCH302 0.1 M NaOH 0.1 M NH3 pH paper turns orange pH = 1 pH = 13 weak acid weak base strong acid pH paper turns blue strong base
Step by Step Solution
3.51 Rating (148 Votes )
There are 3 Steps involved in it
01M HCl strong acid pH1 diagram 2 01M HC2H3O2 Weak acid ... View full answer

Get step-by-step solutions from verified subject matter experts


