Question: Use the template below to construct a MO diagram for the BO molecule and use it to answer the following questions. In a diagram
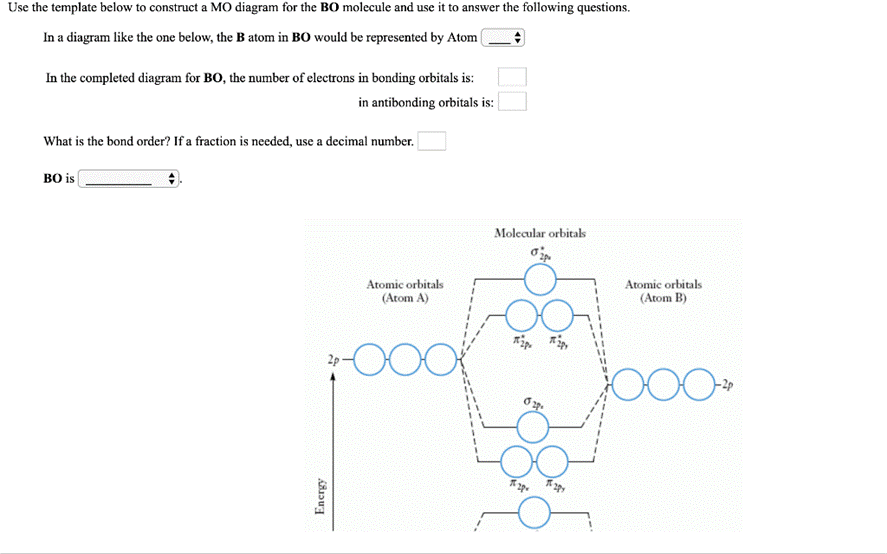
Use the template below to construct a MO diagram for the BO molecule and use it to answer the following questions. In a diagram like the one below, the B atom in BO would be represented by Atom ( In the completed diagram for BO, the number of electrons in bonding orbitals is: in antibonding orbitals is: What is the bond order? If a fraction is needed, use a decimal number. BO is Molecular orbitals Atomic orbitals (Atom A) Atomic orbitals (Atom B) 2p
Step by Step Solution
3.34 Rating (154 Votes )
There are 3 Steps involved in it
B 1s 282 O 18 282 2p4 To faal electro ng z 3 69 k lowen in ... View full answer

Get step-by-step solutions from verified subject matter experts


