Question: Using Eqs. (2.9) and (2.10) for an associated gas and Fig. 2.2, find the compressibility of a 70% specific gravity gas at 5000 psia

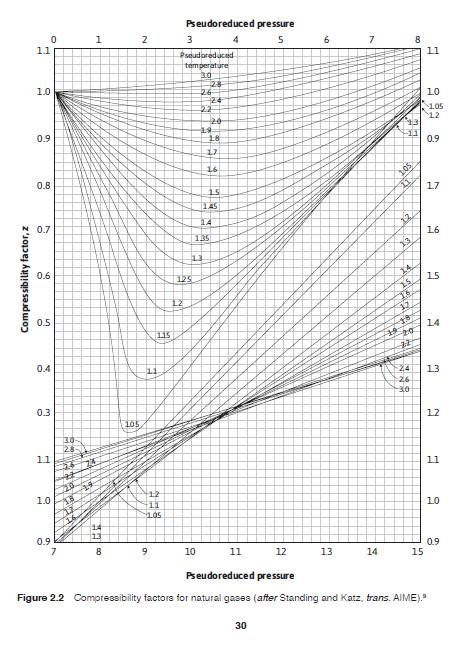
Using Eqs. (2.9) and (2.10) for an associated gas and Fig. 2.2, find the compressibility of a 70% specific gravity gas at 5000 psia and 203F. P = 671.1 + 14.0y - 34.3y? (2.9) pe T = 120.1 + 429Y, 62.9Y? (2.10) pc Pseudoreduced pressure 1 3 4 6. 7 11 11 Pseudoreduced temperature 30 1.0 26 24 22 20 10 1.05 12 0.9 18 0.9 16 0.8 17 15 145 14 0.7 16 135 0.6 15 125 12 0.5 14 115 24 -26 0.4 11 13 0.3 12 105 11 11 12 1.0 10 11 0.9 0.9 15 8 9. 10 11 12 13 14 Pseudoreduced pressure Figure 2.2 Compressibility factora for natural gas88 (after Standing and Katz, trans. AIME). 30 Compressibility factor, z 2.
Step by Step Solution
3.52 Rating (159 Votes )
There are 3 Steps involved in it
ANSWER The compress... View full answer

Get step-by-step solutions from verified subject matter experts


