Using the bond enthalpies in the table below, calculate the enthalpy change (AH) for the reaction...
Fantastic news! We've Found the answer you've been seeking!
Question:
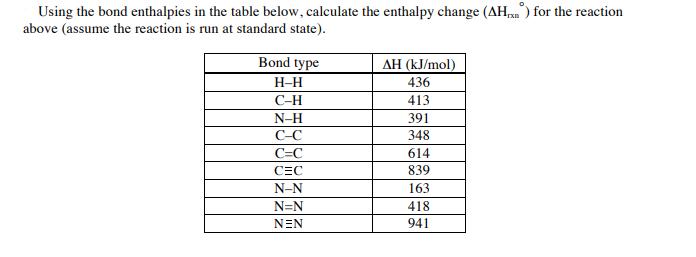
Transcribed Image Text:
Using the bond enthalpies in the table below, calculate the enthalpy change (AH) for the reaction above (assume the reaction is run at standard state). Bond type H-H C-H N-H C-C C=C CEC N-N N=N NEN AH (kJ/mol) 436 413 391 348 614 839 163 418 941 Using the bond enthalpies in the table below, calculate the enthalpy change (AH) for the reaction above (assume the reaction is run at standard state). Bond type H-H C-H N-H C-C C=C CEC N-N N=N NEN AH (kJ/mol) 436 413 391 348 614 839 163 418 941
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Calculate the standard enthalpy change for the reaction 2Al(s) + Fe2O3(s) - 2Fe(s) + Al2O3(s) given that 2Al(s) +3O2(g)- Al2O3(s) n_-1669 - F603(s) .8 mol 2Fe(s) + O2(g) -MR00-822.2 kJ/mol 2(g)
-
Calculate the standard enthalpy change for the reaction of an aqueous solution of barium hydroxide, Ba(OH)2, with an aqueous solution of ammonium nitrate, NH4NO3, at 25oC. (Figure 6.1 illustrated...
-
Calculate the enthalpy change for the reaction P4O6 + 2 O2(g) P4O10(s) given the following enthalpies of reaction: Pals) + 302(g) _ P406(s) P4(s) + 5 Odg)- P4010(s) --1 640.1 kJ --2940.1 kJ
-
Given the monthly returns that follow, find the R2, alpha, and beta of the portfolio. Compute the average return differential with and without sign. Do not round intermediate calculations. Round your...
-
Holden Company produces three products, with costs and selling prices as follows: A particular machine is a bottleneck. On that machine, 3 machine hours are required to produce each unit of Product...
-
The figure below shows 2 tangent circles such that the 9- inch diameter of the smaller circle is equal to the radius of the larger circle. What is the approximate area, in square inches, of the...
-
Terry and Craig share profits in the proportion of one-third and two-thirds respectively. On 1 July 2025, the equity accounts stood as follows. Partners were entitled to 8% interest on capital, and...
-
Dan Aykroyd Corp. was a 30% owner of Steve Martin Company, holding 210,000 shares of Martins common stock on December 31, 2013. The investment account had the following entries. On January 2, 2014,...
-
16. 17. The IUPAC nomenclature of an element with electronic configuration [Rn] 5f46d17s is: (a) Unnibium (b) Unnilunium (c) Unnilquandium (d) Unniltrium The compound(s) that is (are) removed as slag...
-
1. Are you persuaded by the states evidence that it had an individual of a different national origin who was treated similarly to Herawi? If Ward (or other managers) treated everyone equally poorly,...
-
During the last century, production cost structures have significantly changed (see Exhibit 18.1 on text page 18-4). Discuss the reasons for this trend which have resulted in Activity Based Costing...
-
In connection with communication and persuasion, explain the following terms: (a) primacy effect; (b) public commitment; (c) threats and fear; (d) opinion leaders; (e) credibility; and (f) the...
-
Examine the significance of subjective rationality and escalation of commitment in decision making under conditions of uncertainty, with reference to personal investment.
-
Examine the role of leadership in modern teams.
-
Explain the following terms: (a) chunk; (b) schemata and scripts; (c) rehearsal; (d) interference and decay; (e) primacy and recency effects; (f) episodic and generic memory; and (g) imagination in...
-
Discuss the essential features of authentic and servant leadership.
-
The global outbreak of coronavirus (COVID-19) is an unprecedented event that has led to lockdowns and unexpected restrictions on the public as well as the corporate sector across the world. Following...
-
Archangel Corporation prepared the following variance report. Instructions Fill in the appropriate amounts or letters for the question marks in the report. ARCHANGEL CORPORATION Variance...
-
Write molecular and net ionic equations for the successive neutralizations of each of the acidic hydrogens of sulfuric acid with potassium hydroxide. (That is, write equations for the reaction of...
-
The pH of 0.10 M CH3NH2 (methylamine) is 11.8. When the chloride salt of methylamine, CH3NH3Cl, is added to this solution, does the pH increase or decrease? Explain, using Le Chtelier's principle and...
-
Calcium carbonate, a white powder used in toothpastes, antacids, and other preparations, decomposes when heated to about 825oC. What is this temperature in degrees Fahrenheit?
-
Shiloh supplies equipment to the automotive and commercial vehicle markets and other industrial customers. It specializes in materials and designs that reduce vehicle weight and increase fuel...
-
Plaintiffs purchased stock warrants (rights to purchase) for blocks of Osborne Computer Corp., the manufacturer of the first mass-market portable personal computer. Because of inability to produce a...
-
Kronenberger Burgoyne, LLP, was a law firm with two equity partners who agreed to equal ownership as of 2009. Before 2009, Kronenberger had owned a majority interest in the firm, and when, in 2011,...

Study smarter with the SolutionInn App



