Question: Using the nomenclature rules you have learned, assign names to the following molecular compounds. 2nd attempt Part 1 (1 point) Jil See Periodic Table
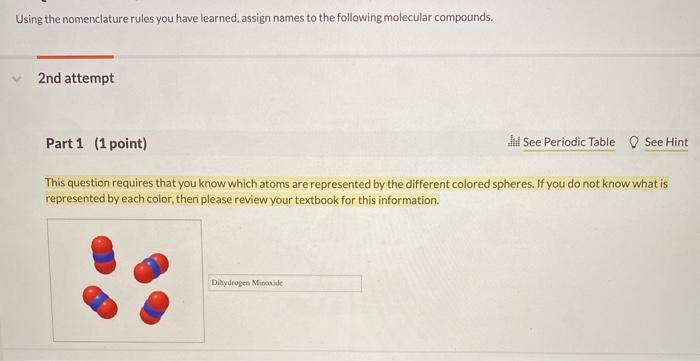

Using the nomenclature rules you have learned, assign names to the following molecular compounds. 2nd attempt Part 1 (1 point) Jil See Periodic Table See Hint This question requires that you know which atoms are represented by the different colored spheres. If you do not know what is represented by each color, then please review your textbook for this information. Dihydrogen Minoxide Part 2 (1 point) Part 3 (1 point) See Hint See Hint
Step by Step Solution
★★★★★
3.43 Rating (159 Votes )
There are 3 Steps involved in it
1 Expert Approved Answer
Step: 1 Unlock

Here red colour represents Oxygen Blue colou... View full answer

Question Has Been Solved by an Expert!
Get step-by-step solutions from verified subject matter experts
Step: 2 Unlock
Step: 3 Unlock


