Question: Using this table, what would be the average tax rate for a single person making $50,000 Taxable Income (dollars) Single Taxpayers Taxable Income (dollars)
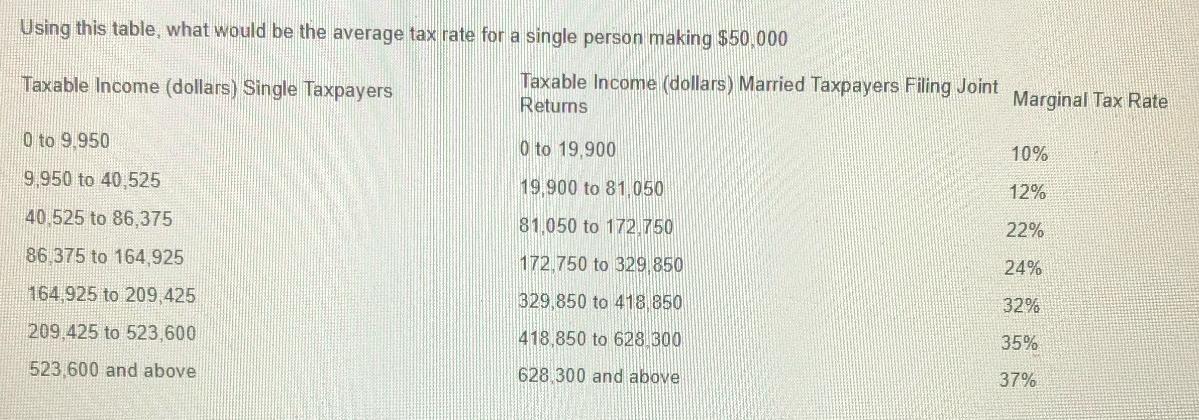
Using this table, what would be the average tax rate for a single person making $50,000 Taxable Income (dollars) Single Taxpayers Taxable Income (dollars) Married Taxpayers Filing Joint Returns Marginal Tax Rate 0 to 9.950 9,950 to 40,525 40,525 to 86,375 0 to 19,900 10% 19,900 to 81,050 12% 81,050 to 172.750 22% 86,375 to 164,925 172,750 to 329,850 24% 164,925 to 209,425 329,850 to 418.850 32% 209,425 to 523,600 418,850 to 628,300 35% 523,600 and above 628,300 and above 37%
Step by Step Solution
3.49 Rating (152 Votes )
There are 3 Steps involved in it
To determine the average tax rate for a single person making 50000 we need to refer to the provid... View full answer

Get step-by-step solutions from verified subject matter experts


