Show your work for your calculations. Mass of crucible + lid Mass crucible + lid+ hydrate...
Fantastic news! We've Found the answer you've been seeking!
Question:
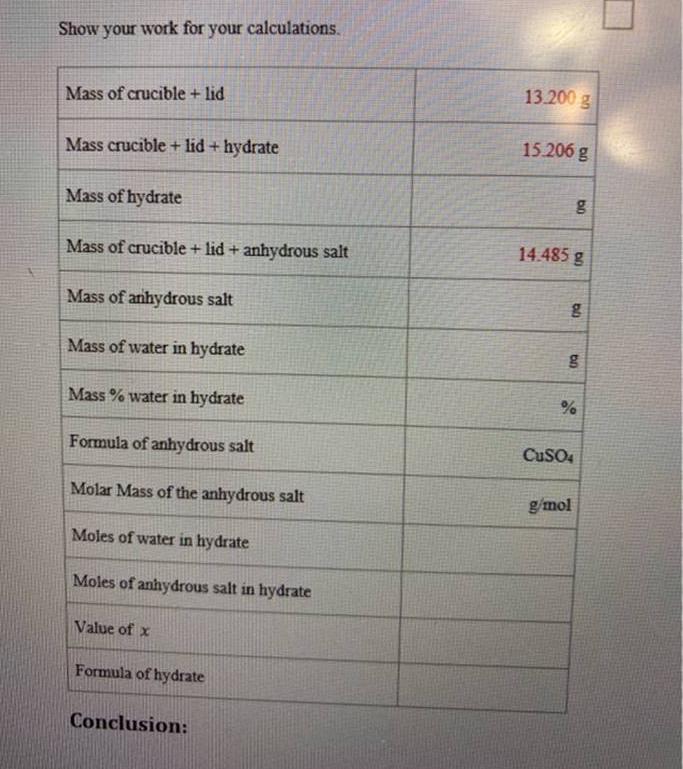

Transcribed Image Text:
Show your work for your calculations. Mass of crucible + lid Mass crucible + lid+ hydrate Mass of hydrate Mass of crucible + lid + anhydrous salt Mass of anhydrous salt Mass of water in hydrate Mass % water in hydrate Formula of anhydrous salt Molar Mass of the anhydrous salt Moles of water in hydrate Moles of anhydrous salt in hydrate Value of x Formula of hydrate Conclusion: 13.200 g 15.206 g bo 14.485 g g g % CuSO4 g/mol Questions: 1. True or False: The hydrate is the salt that remains after heating. 2. True or False: The anhydrous salt does not contain water. 3. The hydrate salt was heated and the weight after heating was determined to be 3.894. Is this the mass of the hydrate salt or of the anhydrous salt? 4. Given the following grams of the substances, what is the percentage of C in the compound? • Mass of C = 2.402 g Mass of the compound = 10.304 g 5. Given the following grams of the elements, what is the empirical formula of the compound? • 2.45 g Si 12.4 g Cl Show your work for your calculations. Mass of crucible + lid Mass crucible + lid+ hydrate Mass of hydrate Mass of crucible + lid + anhydrous salt Mass of anhydrous salt Mass of water in hydrate Mass % water in hydrate Formula of anhydrous salt Molar Mass of the anhydrous salt Moles of water in hydrate Moles of anhydrous salt in hydrate Value of x Formula of hydrate Conclusion: 13.200 g 15.206 g bo 14.485 g g g % CuSO4 g/mol Questions: 1. True or False: The hydrate is the salt that remains after heating. 2. True or False: The anhydrous salt does not contain water. 3. The hydrate salt was heated and the weight after heating was determined to be 3.894. Is this the mass of the hydrate salt or of the anhydrous salt? 4. Given the following grams of the substances, what is the percentage of C in the compound? • Mass of C = 2.402 g Mass of the compound = 10.304 g 5. Given the following grams of the elements, what is the empirical formula of the compound? • 2.45 g Si 12.4 g Cl
Expert Answer:
Answer rating: 100% (QA)
Here are the stepbystep calculations with explanations Mass of crucible lid 13200 g Mass crucible li... View the full answer

Related Book For 

University Physics with Modern Physics
ISBN: 978-0321501219
12th Edition
Authors: Hugh D. Young, Roger A. Freedman, Lewis Ford
Posted Date:
Students also viewed these accounting questions
-
A certain magnesium sulfate salt hydrate sample has a mass of 246.506 mg. After heating and cooling the sample has a new mass of 120.366 mg. How many moles of water were included in 1 mole of the...
-
A solution of 1.00 g of anhydrous aluminum chloride, AlCl3, in 50.0 g of water freezes at 21.11C. Does the molar mass determined from this freezing point agree with that calculated from the formula?...
-
Water of mass m = 20 g is enclosed in a thermally insulated cylinder at the temperature of 0 C under a weightless piston whose area is S = 410 cm2 . The outside pressure is equal to standard...
-
Historically, investment spending has experienced more extreme upward and downward swings than consumer spending. Why do you think this is so?
-
What would the profit margin for the year based on 13-13?
-
Discuss how economic, global, and ethical environments interact with respect to an organization like Mercy Corps?
-
Do the assumptions for Bernoulli trials appear to hold? Explain. If the assumptions hold, identify success and the probability of interest. (a) A TV ratings company will use their electronic...
-
Listed below are altitudes (thousands of feet) and outside air temperatures (degrees Fahrenheit) recorded by the author during Delta Flight 1053 from New Orleans to Atlanta. Is there sufficient...
-
Sing Ltd is investing $600 million in a new manufacturing facility which will increase their capacity by 75%. The resultant present value of future net cash flows is estimated to be $850 million....
-
An FI has purchased a $200 million cap of 9 percent at a premium of 0.65 percent of face value. A $200 million floor of 4 percent is also available at a premium of 0.69 percent of face value. a. If...
-
Show that if E1 and E2 are measurable then m(E1 E2) + m(E1 E2) = m(E1) + m(E2)
-
A pole light is mounted 4.8m above a walkway. The light source uniformly emits 8055 Lumens. (a) Calculate the light level on the pathway directly underneath the light fixture (b) Calculate the light...
-
ypes (s) You are designing a rudimentary pinball machine, and you want your targets to activate whenever the ball pushes the spring back. The target naturally rests at So 1.37 m. Your targets have a...
-
The Chani Company had the following sales forecast: January 440 February 500 March 380 April 300 The company buys products for $320 each and then resells them for $430 each and pays for operating...
-
A small boat is anchored in a lake, and waves are passing by, causing the boat to bob up and down. You observe that when the wave frequency increases, the boat's vertical motion changes. Calculate...
-
A resistor with color code yellow, brown, black, gold measures 42.6 2 on the multimeter. What is the coded resistance? What is the tolerance range? Is this resistor within tolerance? (6 pts.) Rm=...
-
The door company produces four different doors: A, B, C and D. The sales proceeds and costs of these doors are presented below. Sales Revenue Fixed Expenses Variable Expenses Profit/ Loss A B 1.400...
-
Select a mass spectrometric technique with the highest mass resolution for identifying an unknown compound being eluted from a liquid chromatography column
-
A sled starts from rest at the top of a hill and slides down with a constant acceleration. At some later time it is 14.4 m from the top; 2.00 s after that it is 25.6 m from the top, 2.00s later 40.0...
-
Certain rifles can fire a bullet with a speed of 965 m/s just as it leaves the muzzle (this speed is called the muzzle velocity). If the muzzle is 70.0 cm long and if the bullet is accelerated...
-
The two vectors A and B are drawn from a common point, and C = 1 + B. (a) Show that if C2 = A2 + B2, the angle between the vectors A and B is 90. (b) Show that if C2 < A2 + B2, the angle between the...
-
Jupiter's is considering an investment in time and administrative expense on an effort that promises one large payoff in the future, followed by additional expenses over a 10-year horizon. The cash...
-
A large company has the opportunity to select one of seven projects-A, B, C, D, E, F, G-or choose the null (donothing) alternative. Each project requires a single initial investment as shown in the...
-
Management is considering three alternatives to satisfy an urgent need. Each of the alternatives will completely satisfy the need, so no combinations have to be considered. The first costs, operating...

Study smarter with the SolutionInn App


