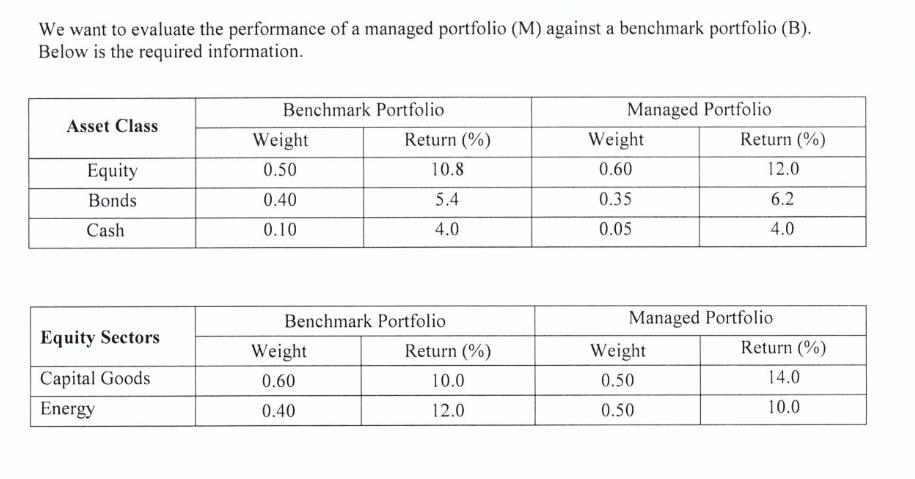
We want to evaluate the performance of a managed portfolio (M) against a benchmark portfolio (B)....
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
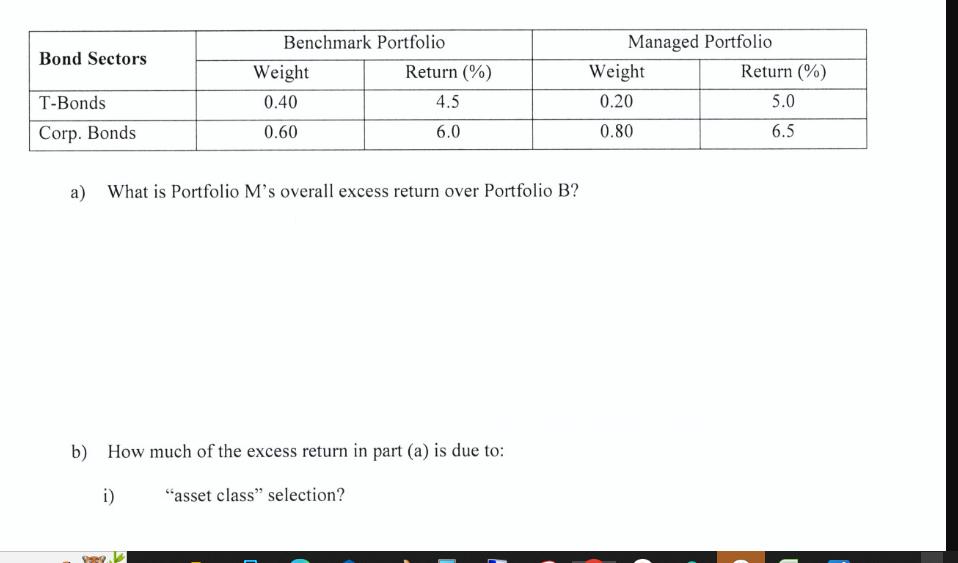
We want to evaluate the performance of a managed portfolio (M) against a benchmark portfolio (B). Below is the required information. Asset Class Equity Bonds Cash Equity Sectors Capital Goods Energy Benchmark Portfolio Weight 0.50 0.40 0.10 Return (%) 10.8 5.4 4.0 Benchmark Portfolio Weight 0.60 0.40 Return (%) 10.0 12.0 Managed Portfolio Weight 0.60 0.35 0.05 Return (%) 12.0 6.2 4.0 Managed Portfolio Weight 0.50 0.50 Return (%) 14.0 10.0 Benchmark Portfolio Managed Portfolio Bond Sectors Weight Return (%) Weight Return (%) T-Bonds 0.40 4.5 0.20 5.0 Corp. Bonds 0.60 6.0 0.80 6.5 a) What is Portfolio M's overall excess return over Portfolio B? b) How much of the excess return in part (a) is due to: i) "asset class" selection? We want to evaluate the performance of a managed portfolio (M) against a benchmark portfolio (B). Below is the required information. Asset Class Equity Bonds Cash Equity Sectors Capital Goods Energy Benchmark Portfolio Weight 0.50 0.40 0.10 Return (%) 10.8 5.4 4.0 Benchmark Portfolio Weight 0.60 0.40 Return (%) 10.0 12.0 Managed Portfolio Weight 0.60 0.35 0.05 Return (%) 12.0 6.2 4.0 Managed Portfolio Weight 0.50 0.50 Return (%) 14.0 10.0 Benchmark Portfolio Managed Portfolio Bond Sectors Weight Return (%) Weight Return (%) T-Bonds 0.40 4.5 0.20 5.0 Corp. Bonds 0.60 6.0 0.80 6.5 a) What is Portfolio M's overall excess return over Portfolio B? b) How much of the excess return in part (a) is due to: i) "asset class" selection?
Expert Answer:
Answer rating: 100% (QA)
Okay here are the steps to solve this a Portfolio Ms overall excess r... View the full answer

Related Book For 

Investment Analysis and Portfolio Management
ISBN: 978-0538482387
10th Edition
Authors: Frank K. Reilly, Keith C. Brown
Posted Date:
Students also viewed these finance questions
-
Are ethnic background and opinion on this issue related? Do a 5 % test. Use the following information to answer question. A questionnaire was sent to 200 students on the campus, asking them to...
-
Are AfricanAmericans opinions equally split among the three categories? Do a 5 % test. Use the following information to answer question. A questionnaire was sent to 200 students on the campus, asking...
-
Four financial analysts were asked to predict earnings growth over the coming year for five oil companies. Their forecasts, as projected percentage increases in earnings, are given in the...
-
Each of the systems in Problems 11 through 18 has a single critical point (x 0 , y 0 ). Apply Theorem 2 to classify this critical point as to type and stability. Verify your conclusion by using a...
-
The budget committee of Urbina Company has collected the following data for its Westwood Store in preparing budgeted income statements for July and August 2012. 1. Expected sales: July $400,000,...
-
Why can you never really have 100% confidence of correctly estimating the population characteristic of interest? Discuss in detail.
-
Show that during the early part of the electron-positron annihilation era, the ratio of the electron number density to the photon number density scaled with temperature as \[\frac{n_{-}}{n_{\gamma}}...
-
The December cash records of Duffy Insurance follow: Duffys Cash account shows a balance of $17,050 at December 31. On December 31, Duffy Insurance received the following bank statement: Additional...
-
Briefly describe TWO (2) drawbacks of NoSQL in comparison with RDBMS ?
-
What is your stance on the efficacy and desirability of community policing as a policy approach? Please elucidate the rationale behind your position, considering its potential impacts on crime...
-
What does the authorization policy provide on Cisco ISE? Associates rule to a specific user Associates rule to a specific device Authenticates a user on the Cisco ISE Authenticates the devices and...
-
A pendulum consists of a bob with mass 5 0 g and a string with length 3 0 cm . It is released from rest from an angle of 1 0 to the vertical. What is the maximum speed of the bob?
-
What role does role-based access control (RBAC) play in enterprise-grade user permissions management, enabling administrators to define access policies based on user roles, job functions, and...
-
How do advanced network security mechanisms, such as Intrusion Detection Systems (IDS), Intrusion Prevention Systems (IPS), and Next-Generation Firewalls (NGFW), employ techniques such as anomaly...
-
Below are the total costs at different levels of output for Merkley Corporation: Production Cost X Cost Y Cost Z 9,600 units $13,500 $11,200 $33,600 8,300 units $11,875 $11,200 $29,050 What type of...
-
Using Alignment Chart, determine the effective length factors K for columns EF, FG. W12 x 50 and KL. Assume WI8 x 60 WIkx 60 %3D 12 ft W12 x 72 W12x58 W24x103 W24x103 unbraced frame (sidesway...
-
Consider the sections of two circuits illustrated above. Select True or False for all statements.After connecting a and b to a battery, the voltage across R1 always equals the voltage across R2.Rcd...
-
a. Given the data in Problem 1, construct an equal-weighted index by assuming $1,000 is invested in each stock. What is the percentage change in wealth for this portfolio? b. Compute the percentage...
-
Draw a hypothetical graph of an efficient frontier of U.S. common stocks. On the same graph, draw an efficient frontier assuming the inclusion of U.S. bonds as well. Finally, on the same graph, draw...
-
It is March 9, and you have just entered into a short position in a soybean meal futures contract. The contract expires on July 9 and calls for the delivery of 100 tons of soybean meal. Further,...
-
When nitrogen and fluorine combine to form a molecule, the most likely chemical formula is: (a) N 3 F. (b)N 2 F. (c) NF 4 . (d) NF. (e) NF 3 .
-
In terms of the periodic table, is there an abrupt or gradual change between ionic and covalent bonds? (a) An abrupt change occurs across the metalloids. (b) Actually, any element of the periodic...
-
A substance consisting of which of the two molecules shown below should have a higher boiling point? Why? (a) the molecule on the left, SCO, because it comes later in the periodic table (b) the...

Study smarter with the SolutionInn App


