Weight of empty 125 ml Erlenmeyer flask = 88.9 g Weight of Erlenmeyer flask with water...
Fantastic news! We've Found the answer you've been seeking!
Question:
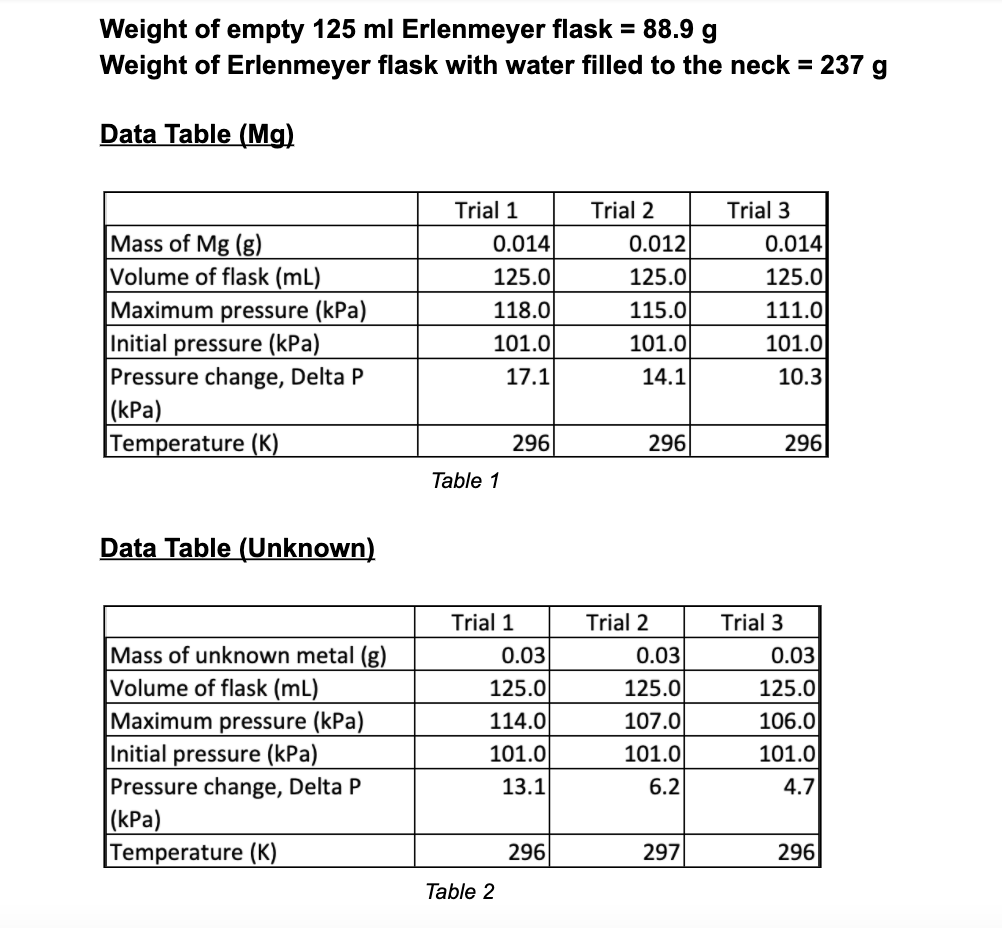


Transcribed Image Text:
Weight of empty 125 ml Erlenmeyer flask = 88.9 g Weight of Erlenmeyer flask with water filled to the neck = 237 g Data Table (Mg) Mass of Mg (g) Volume of flask (ml) Maximum pressure (kPa) Initial pressure (kPa) Pressure change, Delta P (kPa) Temperature (K) Data Table (Unknown) Mass of unknown metal (g) Volume of flask (ml) Maximum pressure (kPa) Initial pressure (kPa) Pressure change, Delta P (kPa) Temperature (K) Trial 1 0.014 125.0 118.0 101.0 17.1 Table 1 296 Trial 1 0.03 125.0 114.0 101.0 13.1 Table 2 296 Trial 2 0.012 125.0 115.0 101.0 14.1 Trial 2 296 0.03 125.0 107.0 101.0 6.2 297 Trial 3 0.014 125.0 111.0 101.0 10.3 296 Trial 3 0.03 125.0 106.0 101.0 4.7 296 Data Analysis: 1. Using the measured weight of the empty 125 mL weight when filled with water, and the known density of water at the temperature of the measurement, calculate the "effective" volume of the 125 mL Erlenmeyer flask, i.e., the volume available for occupancy by the H₂ gas. (You must also take into account the volume of the added HCl solution, which decreases the volume available to the H2 gas.) 2. Calculate the molar amounts of each piece of magnesium and unknown metal that you used. ENEW 3. Based on the measured temperatures and pressure changes for each reaction, use the ideal gas the molar amount of hydrogen gas that was produced in each reaction. 4. Calculate the volume occupied by one mole of hydrogen gas (molar volume) at STP. 5. Compare your calculated molar volume, at STP, with the accepted molar volume of an ideal gas at STP, viz., 22.41 L/mol. If the values do not compare well, suggest possible sources of experimental error. 6. Use your calculated value for the STP molar volume to determine the atomic weight of your unknown metal. What is the likely identity of the metal? Weight of empty 125 ml Erlenmeyer flask = 88.9 g Weight of Erlenmeyer flask with water filled to the neck = 237 g Data Table (Mg) Mass of Mg (g) Volume of flask (ml) Maximum pressure (kPa) Initial pressure (kPa) Pressure change, Delta P (kPa) Temperature (K) Data Table (Unknown) Mass of unknown metal (g) Volume of flask (ml) Maximum pressure (kPa) Initial pressure (kPa) Pressure change, Delta P (kPa) Temperature (K) Trial 1 0.014 125.0 118.0 101.0 17.1 Table 1 296 Trial 1 0.03 125.0 114.0 101.0 13.1 Table 2 296 Trial 2 0.012 125.0 115.0 101.0 14.1 Trial 2 296 0.03 125.0 107.0 101.0 6.2 297 Trial 3 0.014 125.0 111.0 101.0 10.3 296 Trial 3 0.03 125.0 106.0 101.0 4.7 296 Data Analysis: 1. Using the measured weight of the empty 125 mL weight when filled with water, and the known density of water at the temperature of the measurement, calculate the "effective" volume of the 125 mL Erlenmeyer flask, i.e., the volume available for occupancy by the H₂ gas. (You must also take into account the volume of the added HCl solution, which decreases the volume available to the H2 gas.) 2. Calculate the molar amounts of each piece of magnesium and unknown metal that you used. ENEW 3. Based on the measured temperatures and pressure changes for each reaction, use the ideal gas the molar amount of hydrogen gas that was produced in each reaction. 4. Calculate the volume occupied by one mole of hydrogen gas (molar volume) at STP. 5. Compare your calculated molar volume, at STP, with the accepted molar volume of an ideal gas at STP, viz., 22.41 L/mol. If the values do not compare well, suggest possible sources of experimental error. 6. Use your calculated value for the STP molar volume to determine the atomic weight of your unknown metal. What is the likely identity of the metal?
Expert Answer:
Answer rating: 100% (QA)
Experiment 1 Mg Trial 1 Mass of Mg 0014 g Maximum pressur... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Water enters a pump at 100 kPa with a mass flow rate of 20 kg/s and exits at 500 kPa with the same mass flow rate. If the density () of water is 1000 kg/m3, determine (a) the net rate of flow work...
-
The mass of the Empty Flask is 49.6536gand the Density is assumed to be 1. Ill fipet Tlalibiration. True Volume Appeck ent alPpRalent Mass of mos5 of pipet scadig (m1) 0,00 1000 Plask (5) 4A 6536...
-
Measured data for pressure versus volume during the compression of a refrigerant within the cylinder of a refrigeration compressor are given in the table below. Using data from the table complete the...
-
An air-filled X-band rectangular waveguide has dimensions a = 2.286 cm and b = 1.016 cm. If the waveguide has copper walls ( = o , = o , = 5.8 x 10 7 S/m), find the attenuation in dB/m due to the...
-
The Faraway Moving Company is involved in a major plant expansion that involves the expenditure of $200 million in the coming year. The firm plans on financing the expansion through the retention of...
-
As seen in Figure, a square loop of wire has sides of length 2.0 cm. A magnetic field is directed out of the page; its magnitude is given by B = 4.0t2y, where B is in teslas, t is in seconds, and y...
-
Larry Riley formed a rental agreement for a self-storage unit with Iron Gate Self Storage in December 2003. The agreement stated that a maximum of \($5,000\) of personal property could be stored in...
-
The book balance in the checking account of Johnson Enterprises as of October 31 is $5,718. The bank statement shows an ending balance of $5,217. The following information is discovered by (1)...
-
produce a reflective comparison of China and the U.S. in terms of social/political/economic systems. Given the focus of this course and this particular module, what is the fundamental basis for the...
-
Allen Furniture is a manufacturer of hand-crafted furniture. At the start of January, Allen employs 20 trained craftspeople. They have forecasted their labor needs over the next 12 months as shown in...
-
A certain radioactive isotope is a by - product of some nuclear reactors. Due to an explosion, a nuclear reactor experiences a massive leak of this radioactive isotope. Fortunately, the isotope has a...
-
How could producing urease support the survival of a bacterium colonizing the stomach, such as Helicobacter pylori?
-
During the current period, JT Engineering experienced a holding gain on one of its securities. This change in the security's value was reported as part of net income. Based on this information, what...
-
During Tax Year 2022, Lalita purchased a new machine for her business for $30,000. She uses the machine exclusively for business. Her business income is $145,000. She elects to deduct the property...
-
What is investment banking ? What role does the investment banking industry play in society and in finance more specifically? What are some of the key investment banking activities? What are some of...
-
Explain what adaptation of a sensory receptor is and why it is important. Are there some receptors that don't adapt? Why might it be important that these receptors don't adapt?
-
The big pen store supplies pens to the public. The following transactions took place during the month of May 2021: 14 May: Sold goods on account to Susie's pen store for $3,600, terms 2/10, n/30. The...
-
Grace is training to be an airplane pilot and must complete five days of flying training in October with at least one day of rest between trainings. How many ways can Grace schedule her flying...
-
When 0.273 g of Mg is heated strongly in a nitrogen (N2) atmosphere, a chemical reaction occurs. The product of the reaction weighs 0.378 g. Calculate the empirical formula of the compound containing...
-
Greenhouse gases absorb (and trap) outgoing infared radiation (heat) from Earth and contribute to global warming. The molecule of a greenhouse gas either possesses a permanent dipole moment or has a...
-
Match each of the elements on the right with its description on the left: (a) A pale yellow gas Nitrogen (N2) that reacts with water. Boron (B) (b) A soft metal that reacts Aluminum (Al) with water...
-
The business staff of the law firm Frampton, Davis & Smythe has constructed the following report that breaks down the firms overall results for last month into two business segmentsfamily law and...
-
The Excel worksheet form that appears below is to be used to recreate portions of Review Problem 1 relating to Dexter Corporation. Download the workbook containing this form from Connect, where you...
-
Millard Corporation is a wholesale distributor of office products. It purchases office products from manufacturers and distributes them in the West, Central, and East regions. Each of these regions...

Study smarter with the SolutionInn App


