What fraction of the lactic acid/lactate mixture is lactic acid? Print Question 19 of 20 Calculator Incorrect
Fantastic news! We've Found the answer you've been seeking!
Question:
What fraction of the lactic acid/lactate mixture is lactic acid?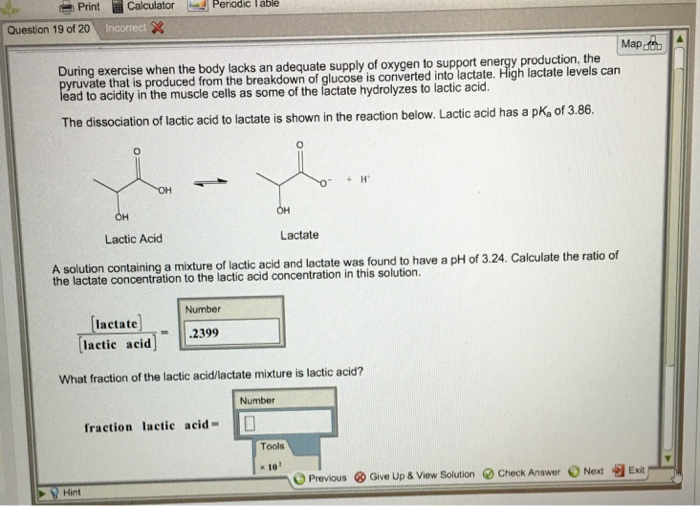

Transcribed Image Text:
Print Question 19 of 20 Calculator Incorrect Map During exercise when the body lacks an adequate supply of oxygen to support energy production, the pyruvate that is produced from the breakdown of glucose is converted into lactate. High lactate levels can lead to acidity in the muscle cells as some of the lactate hydrolyzes to lactic acid. The dissociation of lactic acid to lactate is shown in the reaction below. Lactic acid has a pK₂ of 3.86. Hint OH Periodic Table OH Lactic Acid Number 2399 A solution containing a mixture of lactic acid and lactate was found to have a pH of 3.24. Calculate the ratio of the lactate concentration to the lactic acid concentration in this solution. OH fraction lactic acid- Lactate [lactate] [lactic acid] What fraction of the lactic acid/lactate mixture is lactic acid? Number H Tools * 10¹ Previous Give Up & View Solution Check Answer Next Exit Print Question 19 of 20 Calculator Incorrect Map During exercise when the body lacks an adequate supply of oxygen to support energy production, the pyruvate that is produced from the breakdown of glucose is converted into lactate. High lactate levels can lead to acidity in the muscle cells as some of the lactate hydrolyzes to lactic acid. The dissociation of lactic acid to lactate is shown in the reaction below. Lactic acid has a pK₂ of 3.86. Hint OH Periodic Table OH Lactic Acid Number 2399 A solution containing a mixture of lactic acid and lactate was found to have a pH of 3.24. Calculate the ratio of the lactate concentration to the lactic acid concentration in this solution. OH fraction lactic acid- Lactate [lactate] [lactic acid] What fraction of the lactic acid/lactate mixture is lactic acid? Number H Tools * 10¹ Previous Give Up & View Solution Check Answer Next Exit
Expert Answer:
Answer rating: 100% (QA)
This problem is based on the concept of the HendersonHasselbalch equation The HendersonHasselbalch e... View the full answer

Related Book For 

Thermodynamics An Engineering Approach
ISBN: 978-0073398174
8th edition
Authors: Yunus A. Cengel, Michael A. Boles
Posted Date:
Students also viewed these chemistry questions
-
What fraction of the radioactive cobalt nuclei whose half-life is 71.3 days decays during a month?
-
What fraction of the total kinetic energy is rotational for the following objects rolling without slipping on a horizontal surface? (a) A uniform solid cylinder; (b) A uniform sphere; (c) A...
-
What fraction of the light shining straight at a piece of clear glass is reflected from the first surface?
-
Suppose you have a subject property with a 105,000 sq. ft. lot and existing improvements for which you estimate the reproduction cost new to be $2,500,000, physical deterioration to be $400,000,...
-
The president of Mexico has asked you for advice on the likely economic consequences of the following five policies designed to improve Mexicos economic environment. Describe the consequences of each...
-
Find the average production cycle for Rian Company. Use the below information for solving the question 2008 Selected Income Statement Items for Rian Company Cash Sales ..... $ 298,000 Credit Sales...
-
In 1951, DuPont began using the chemical perfluorooctanoic acid to manufacture Teflon. Due to the dangerous nature of the chemical, DuPont was given special instructions by its supplier to dispose of...
-
Greene Corporation has four divisions: the assembly division, the processing division, the machining division, and the packing division. All four divisions are under the control of the vice president...
-
A government institutes a rule that schools are allowed to sell whatever foods it chooses, provided that those foods are not high in sugar. Such a ruling is aligned with which economic system?
-
Using the check register below and the following bank statement, prepare a bank reconciliation for Lee.com. Note: Input all amounts as positive values. Round your answers to 2 decimal places. NUMBER...
-
Derive the capacitance for a)a parallel plate capacitor with cross sectional area A and plate separation d b)a cylindrical capacitor with inner conductor radius a and outer conductor radius b and...
-
Identify and briefly define 1 of the following concepts: Opportunity cost Sunk cost Costbenefit analysis Based on the concept that you have selected, respond to each of the following: What are the...
-
er the last 60 years, the frozen food market has boomed. In 2012, sales were $44 billion (about $140 per person in the US), about 44 times what they were in the mid-1950s. Increased labor force...
-
A t-shirt is launched at an angle of 63.6 at 25.8 m/s. The shirt is launched at a person in the stands a horizontal distance of 30.6 m away and 27.7 m above the ground. How many meters will the...
-
The low demand in late November 2018 could be due to various factors. One possibility is that it's related to the Thanksgiving holiday, which typically falls in late November. Many people travel out...
-
In Florida, and was reported in the Wall Street Journal in 2002. First, get out a piece of paper and draw a circle in the middle of it about the size of an apple, draw a second circle just slightly...
-
Snead currently produces 120,000 units at a cost of $400,000. Of the cost of $400,000, $200,000 is a fixed cost. Next year Snead expects to produce 145,000 units. By Snead relevant range for...
-
You are maintaining a subsidiary ledger account for Police-Training Expenditures for 2013. The following columns are used: Inventory purchases are initially recorded as expenditures. Record the...
-
Determine the maximum amount of load, in kg, the balloon described in Prob. 1-102 can carry.
-
Methyl alcohol (CH3OH) is burned with 100 percent excess air. During the combustion process, 60 percent of the carbon in the fuel is converted to CO2 and 40 percent is converted to CO. Write the...
-
Estimate the sfg of the substance in Problem 12-25E at 15oF. Problem 12-25E 0.5-lbm of a saturated vapor is converted to a saturated liquid by being cooled in a weighted piston- cylinder device...
-
A running mountain lion can make a leap 10.0 m long, reaching a maximum height of 3.0 m. a. What is the speed of the mountain lion just as it leaves the ground? b. At what angle does it leave the...
-
The moon completes one (circular) orbit of the earth in 27.3 days. The distance from the earth to the moon is 3.84 10 8 m. What is the moons centripetal acceleration?
-
A soccer player takes a free kick from a spot that is 20 m from the goal. The ball leaves his foot at an angle of 32, and it eventually hits the crossbar of the goal, which is 2.4 m from the ground....

Study smarter with the SolutionInn App


