What gradient series over periods [1,5] is equivalent at 16% to a uniform series with A...
Fantastic news! We've Found the answer you've been seeking!
Question:
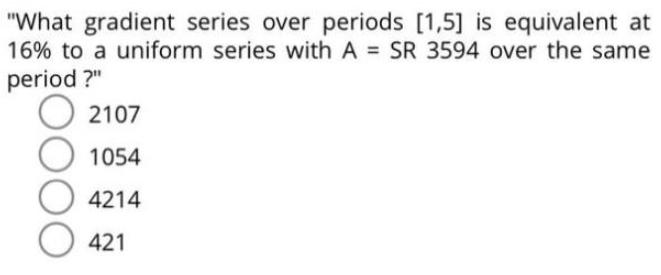
Transcribed Image Text:
"What gradient series over periods [1,5] is equivalent at 16% to a uniform series with A = SR 3594 over the same period?" 2107 1054 4214 421 "What gradient series over periods [1,5] is equivalent at 16% to a uniform series with A = SR 3594 over the same period?" 2107 1054 4214 421
Expert Answer:
Answer rating: 100% (QA)
The answer is 421 Explanation A uniform series with A SR 3594 over the period 15 is equ... View the full answer

Related Book For 

Fundamentals of Thermodynamics
ISBN: 978-0471152323
6th edition
Authors: Richard E. Sonntag, Claus Borgnakke, Gordon J. Van Wylen
Posted Date:
Students also viewed these accounting questions
-
What uniform series of cash flows is equivalent to a $15,000 cash flow occurring today if the uniform series of cash flows occur at the end of each year for the next five years and the periodic...
-
What uniform series of cash flows is equivalent to a $100,000 cash flow, ten years from now, if the uniform cash flows occur at the end of the year for the next ten years and the periodic interest...
-
What uniform series of cash flows is equivalent to a $100,000 cash flow, fifteen years from now, if the uniform cash flows occur at the end of the year for the next fifteen years and the periodic...
-
Why will a reduction in the real interest rate increase investment spending, other things equal?
-
Suppose you manage a Coffee Plus! restaurant. Identify the missing amount for eachsituation: Total Assets Total Liabilities Stockholders' Equity $340,000 356,000 + $320,000 57,000 b. 135,000 401,000
-
Two identical batteries, each with an emf E and an internal resistance r, can be connected across a resistance R either in series or in parallel. Is the power supplied to R greater when R < r or when...
-
Event E: rolling a number divisible by 4 A probability experiment consists of rolling a 12-sided die, numbered 1 to 12. Find the probability of the event.
-
Sarah Johnson was a trusted employee of Evergreen Trust Bank. She was involved in everything. She worked as a teller, she accounted for the cash at the other teller windows, and she recorded many of...
-
Ronald Robert has spent much of the last few years gambling and playing soccer in Chicago. He has been largely unemployed during that period. About two years ago, however, he got his act together and...
-
What Happens When You Dont Deliver on Your Promises Web: www.clearly.ca Facebook: Clearly Canadian If a new product or service seems like the perfect option to solve a problem or capitalize on an...
-
During June, Agron Inc. purchases of direct materials totaled $119,000; direct labor for the month was 3,400 hours at $10.00 per hour. Agron also incurred the following overhead costs: utilities,...
-
Suppose you replaced the coil with one that has double the radius. How would that affect the average emf for this same timeline? Explain your reasoning (no calculations needed, but you can use...
-
On March 1, 2025, Cullumber Company acquired real estate, on which it planned to construct a small offic building, by paying $87,000 in cash. An old warehouse on the property was demolished at a cost...
-
Company's December 31 Balance Sheets Assets 2022 2021 Cash $ 72,000 $ 65,000 Accounts receivable 439,000 328,000 Inventories 894,000 813,000 Total current assets $ 1,405,000 $ 1,206,000...
-
4. A driver initially at rest accelerates her 1500 kg car to a final velocity of 30 m/s. a. What is the kinetic energy of the car at her final velocity? b. The force acting on the car is 6000 N. How...
-
A coupon bond pays 8% annually with annual cash flow of $1000 what is the Face value of the coupon bond?
-
In Atlanta, a nonprofit discovered that its director of facilities had embezzled more than $1.1 million. The FBI stated that an employee had submitted invoices for bogus expenses and then deposited...
-
Funds are separate fiscal and accounting entities, each with its own self-balancing set of accounts. The newly established Society for Ethical Teachings maintains two funds-a general fund for...
-
A piston cylinder hAs constant pressure of 2000 kPa with water at 20oC. It is now heated up to 100oC. Find the heat transfer and the entropy change using the steam tables. Repeat the calculation...
-
A water-cooling tower for a power plant cools 45C liquid water by evaporation. The tower receives air at 19.5C, = 30%, 100 kPa that is blown through/over the water such that it leaves the tower at...
-
Two un insulated tanks of equal volume are connected by a valve. One tank contains a gas at a moderate pressure P1, and the other tank is evacuated. The valve is opened and remains open for a long...
-
To cool water for drinking during summer, \(1 \mathrm{~kg}\) of ice at \(-2^{\circ} \mathrm{C}\) is mixed with \(5 \mathrm{~kg}\) of water at \(27^{\circ} \mathrm{C}\) in an insulated container...
-
In a cyclic process, heat transfers are \(+14.7 \mathrm{~kJ},-25.2 \mathrm{~kJ},-3.56 \mathrm{~kJ}\) and \(+31.5 \mathrm{~kJ}\). What is the net work for this cyclic process?
-
A heat pump operates between two identical bodies of specific heat \(C\) at \(T_{1}\). The operation of the pump cools down one of the bodies to \(T_{2}\). Show that for the operation of pump the...

Study smarter with the SolutionInn App


