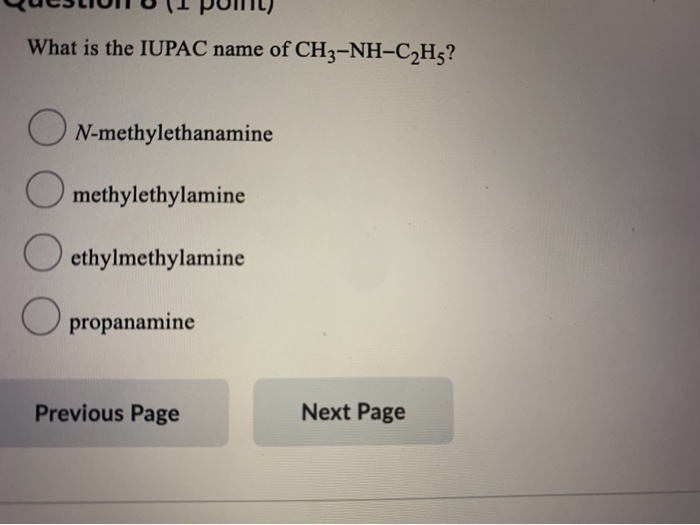
What is the IUPAC name of CH3-NH-CH5? ON-methylethanamine methylethylamine ethylmethylamine propanamine Previous Page Next Page If...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
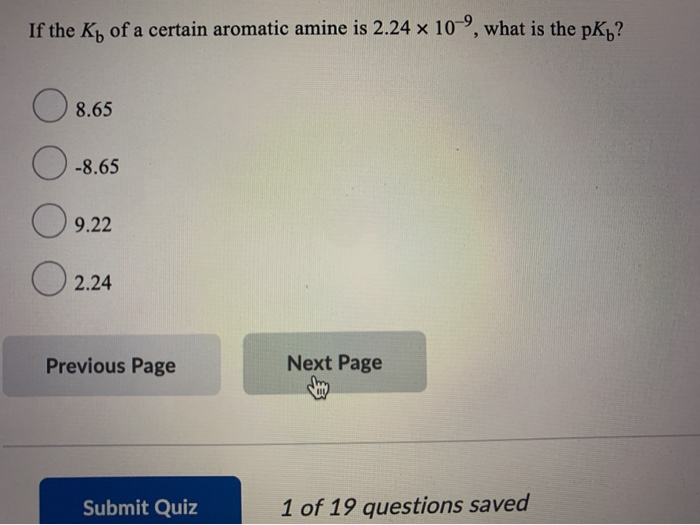
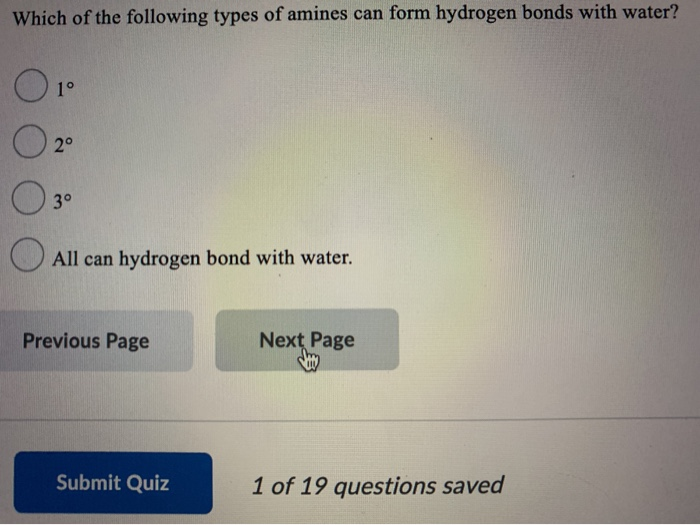
What is the IUPAC name of CH3-NH-C₂H5? ON-methylethanamine methylethylamine ethylmethylamine propanamine Previous Page Next Page If the Kt of a certain aromatic amine is 2.24 x 10, what is the pK₁? 8.65 -8.65 9.22 2.24 Previous Page Submit Quiz Next Page 1 of 19 questions saved Which of the following types of amines can form hydrogen bonds with water? 0 1° O 2⁰ 3° All can hydrogen bond with water. Previous Page Submit Quiz Next Page 1 of 19 questions saved What is the IUPAC name of CH3-NH-C₂H5? ON-methylethanamine methylethylamine ethylmethylamine propanamine Previous Page Next Page If the Kt of a certain aromatic amine is 2.24 x 10, what is the pK₁? 8.65 -8.65 9.22 2.24 Previous Page Submit Quiz Next Page 1 of 19 questions saved Which of the following types of amines can form hydrogen bonds with water? 0 1° O 2⁰ 3° All can hydrogen bond with water. Previous Page Submit Quiz Next Page 1 of 19 questions saved
Expert Answer:
Answer rating: 100% (QA)
Solution The polynomial is going through three data points tv v and t3v3 where from the above table ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
What is the IUPAC name of this polymer? I-U-I I-C-H I-C-H I-U-H I-C-H I-C-H I-C-H I-U-H
-
What is the IUPAC name of the compound shown? (a) (E )-3-Methyl-2-hexenoic acid (b) (Z )-3-Methyl-2-hexenoic acid (c) (E )-3-Methyl-3-hexenoic acid (d) (Z )-3-Methyl-3-hexenoic acid H;C CO.H C=C...
-
What is the IUPAC name of the compound shown in the margin? (a) Isopropyl 2-fluoro3-methylbutanoate; (b) 2-fluoroisobutanoyl 2-propanoate; (c) 1-methylethyl 2-fluorobutyrate; (d) 2-fluoroisopropyl...
-
Under what circumstance is it most appropriate to travel in reverse with a load? A. When you are carrying a non-standard load B. When a tall load obstructs your forward vision C. When the load is...
-
Can a financial institution keep borrowers from engaging in risky activities if there are no restrictive covenants written into the loan agreement?
-
Using the result obtained in Problem 57, show that three distinct points (x 1 , y 1 ), (x 2 , y 2 ) and (x 3 , y 3 ) are collinear (lie on the same line) if and only if 1 1 1 2 2 1 3 0 | 1
-
When testing several hypotheses, it is reasonable to reject every hypothesis for which P < 0.05. In Exercises 5 and 6, determine whether the statement is true or false. If the statement is false,...
-
1. Develop an implementation plan that would be useful to the Training group in changing to a computerized project tracking system. Use a paragraph to explain your approach. Be sure that what you are...
-
Define the term "corporate governance".Is good governance good business globally?Explain.
-
An Engagement supervisor has asked the audit senior to perform the following procedures related to an engagement. For each procedure listed, identify the financial statement assertion being tested by...
-
Suppose that the economy is in a state of equilibrium where Aggregate Demand (AD) equals Aggregate Supply (AS), and the economy is producing at its full employment level. Suppose also that the...
-
In the formula, the weights of autonomy and feedback are how many times the weight of variety, identity, and significance? SPS= SPS= SPS= V1+V2+11+12+S1+S2 6 6 X X X A1+A2 2 2 X X X 11 F1+F2 2 2
-
Use the product rule to find the derivative of the given function. y=(5x+6x) y=
-
Review the following article: Rosenkoetter, M., Nardi, D., & Bowcutt, M. (2017). Internationally educated nurses in transition in the United States: Challenges and mediators. The Journal of...
-
Differentiate the function. y' = y= = (8x-x+1)(-x+4)
-
Angela works at a company that emphasizes competitiveness among its employees. Angela works at a company __________ in __________. a) high; humane orientation b) high; assertiveness c) low;...
-
When two organizations with very political environments interact with one another the political environments between them can hurt performance in collaborative projects. Indicate whether this is true...
-
Arlington Merchants reported the following on its income statement for the fiscal years ending December 31, 2016 and 2015. 2016 2015 Sales $4,857,500 $4,752,900 Cost of goods sold 3,258,950 3,207,000...
-
(a) Predict the direction of reaction when chlorine gas is added to an equilibrium mixture of COCl 2 , CO, and Cl 2 . The reaction is COCl 2 (g) CO(g) + Cl 2 (g) (b) What is the direction of...
-
The n quantum number of an atomic orbital is 6. What are the possible values of l? What are the possible values of ml if the l quantum number is 5?
-
Consider the complex ion [CoF6]3. a. What is the geometry? b. Which is a more likely color for this ion to absorb, red or blue? c. Would you expect this complex to be high or low spin?
-
With your instructor's consent, identify a company and perform a background review of it to identify high-risk areas for an upcoming audit. Utilize all the electronic sources that have information...
-
According to the AICPA's ethical standards, an auditor would be considered in- dependent in which of the following instances? a. The auditor has an automobile loan from a client bank. b. The auditor...
-
Explain how "privileged communication" differs from confidential information.

Study smarter with the SolutionInn App


