What is the oxidation number of chromium in Cr20; ? OA +12 O B. 7 OD.13...
Fantastic news! We've Found the answer you've been seeking!
Question:
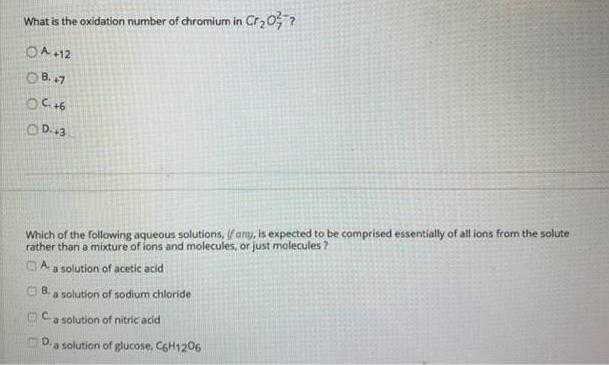
Transcribed Image Text:
What is the oxidation number of chromium in Cr20; ? OA +12 O B. 7 OD.13 Which of the following aqueous solutions, if amy, is expected to be comprised essentially of all ions from the solute rather than a mixture of ions and molecules, or just molecules ? a solution of acetic acid O 8. a solution of sodium chloride OCa solution of nitric acid D.a solution of glucose, CGH1206 What is the oxidation number of chromium in Cr20; ? OA +12 O B. 7 OD.13 Which of the following aqueous solutions, if amy, is expected to be comprised essentially of all ions from the solute rather than a mixture of ions and molecules, or just molecules ? a solution of acetic acid O 8. a solution of sodium chloride OCa solution of nitric acid D.a solution of glucose, CGH1206
Expert Answer:
Answer rating: 100% (QA)
Solution Answer 1 The correct answer is c 6 The sum of the oxidation numbers in Cr 2 O 7 2 a polyato... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
A complex is written as NiBr2.6NH3 (a) What is the oxidation state of the Ni atom in this complex? (b) What is the likely coordination number for the complex? (c) If the complex is treated with...
-
List the different nitrogen oxides. What is the oxidation number of nitrogen in each?
-
Which of the following aqueous solutions has the highest pH and which has the lowest? a. 0.1 M NH3; b. 0.1 M NH4Br; c. 0.1 M NaF; d. 0.1 M NaCl
-
2. C. b. d. e. a. A proposed public project would produce the following pattern of individual benefits and costs. Revise only the cost shares of individuals C and E to meet the Pareto Criterion. (7...
-
What is a project finance (PF) loan? What role does a stand-alone company play in the typical project finance deal?
-
In determining whether a regression models error terms are positively correlated for the model from its least-square residuals. a. State the null and alternative hypothesis for the mentioned...
-
DeLaval turbine is a/an _____.
-
The manager of the Burger Doodle restaurant believes the time to fill orders at the drive-through window is too long. She suspects that the window cashier spends too much time making change, and she...
-
What are the services rendered by Hosted SP? How has the nature of software deployment changed over time? What is the value-added by Hosted SP? STT Reflection: Hosted SP noted difficulty in...
-
Joseph and Diane Smith 1580 West Street Chatham, VA 24531 Joseph and Diane are both 35 and have no dependents. If your clients receive a refund, they want the full amount refunded to them. Diane is...
-
Selected ratios for Pitka Corporation are as follows: 2020 2019 2018 Working capital $10,000 $25,000 $35,000 Current ratio 1.1:1 1.2:1 1.2:1 Debt to total assets 34.2% 42.3% 40.0% Earnings per share...
-
A normal distribution has a mean of 85.7 and a standard deviation of 4.85. Find data values corresponding to the values of z given in Problems 42-45. \(z=-3.46\)
-
A school board wishes to determine opinions of parents regarding the assigning of homework in mathematics classes. Which of the following procedures would be most appropriate for obtaining a...
-
In Problems 7-18, a sample of paired data gives a linear correlation coefficient \(r\). In each case, use Table 14.10 to determine whether there is a significant linear correlation. Table 14. 10...
-
Nonprobability sampling assumes that some elements of the population have no chance of selection or the probability of selection can't be accurately determined. Some types of nonprobability sampling...
-
Draw a scatter diagram and find \(r\) for the data shown in each table in Problems 25-30. x 0 1 2 3 4 y 25 19 16 12 10
-
How would you use call and/or put option to protect the value of your portfolio? The current value of the portfolio is $10 million. The portfolio is made up of $6 million investment in Ford Motor...
-
Illini Company, Inc. Balance Sheet as of 12/31/20X0 Assets Current Assets: Cash $1,500,000 Accounts receivable, net 18,000 Inventory 50,000 Total current assets 1,568,000 Equipment 90,000 Goodwill...
-
Strontium-90, is a radioactive decay product of nuclear fallout from nuclear weapons testing. Because of its chemical similarity to calcium, it is incorporated into the bones if present in food. The...
-
One isotope of a metallic element has mass number 80 and has 55 neutrons in the nucleus. An atomic ion has 23 electrons. Write the symbol for this ion (give the symbol for the nucleus and give the...
-
Phthalic acid, H2C8H4O4, is a diprotic acid used in the synthesis of phenolphthalein indicator. Ka1 = 1.2 103, and Ka2 = 3.9 106.
-
Lennon Industries had the following transactions. 1. Borrowed 5,000 from the bank by signing a note. 2. Paid 3,900 cash for a computer. 3. Purchased 650 of supplies on account. Instructions a....
-
Ong Enterprises had the following selected transactions. 1. Shareholders invested NT\($40,000\) cash in the business in exchange for ordinary shares. 2. Paid office rent of NT\($8,400. 3. Performed...
-
Presented below is the ledger for Shumway Co. (amounts in thousands of ). Instructions a. Reproduce the journal entries for the transactions that occurred on October 1, 10, and 20, and provide...

Study smarter with the SolutionInn App


