What is the product of the following reaction? OI -OH O Q O II O III...
Fantastic news! We've Found the answer you've been seeking!
Question:
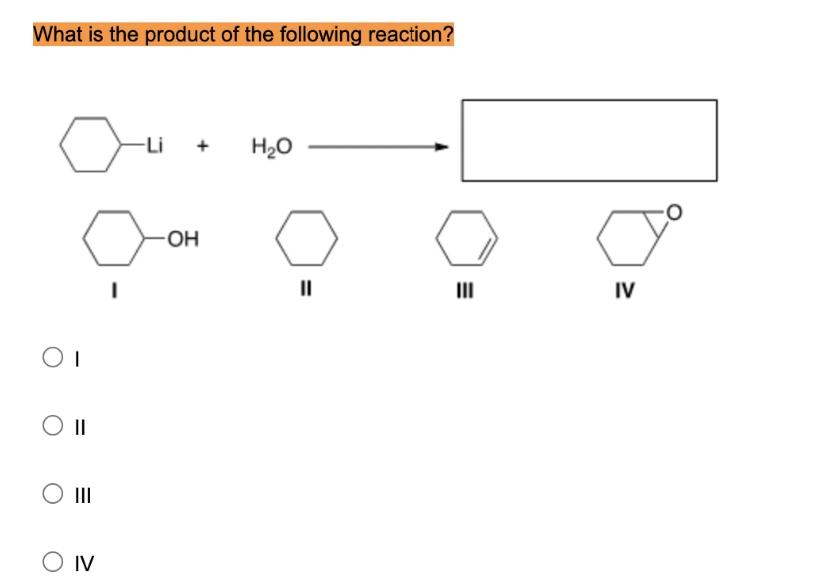
Transcribed Image Text:
What is the product of the following reaction? OI -OH O Q O II O III -Li O IV H₂O IV What is the product of the following reaction? OI -OH O Q O II O III -Li O IV H₂O IV
Expert Answer:
Answer rating: 100% (QA)
II cyclohexane Here is the handwritten explanation wit... View the full answer

Related Book For 

Organic Chemistry
ISBN: 978-1118133576
11th edition
Authors: Graham Solomons, Craig Fryhle, Scott Snyder
Posted Date:
Students also viewed these chemistry questions
-
What is the product of the following SuzukiMiyaura coupling? Br B(OR)2 + Pd catalyst Base
-
What is the product of the following reaction? A) I B) II C) III D) IV NH2 (2 equivalents) I =c-c-CH;CH, H H =c-C-CHCH, HH CEC-CH,CH C=C-CH,CH, IV
-
What is the product of the following reaction? A) I B) II C) III D) IV NH2 (2 equivalents) I =c-c-CH;CH, H H =c-C-CHCH, HH CEC-CH,CH C=C-CH,CH, IV
-
Consider the no-trade input/output situation presented in the following table for countries X and Y. Assuming that free trade is allowed, develop a scenario that will benefit the citizens of...
-
Garys Great Cars purchases high-performance auto parts from a Nebraska vendor. Dave Simon, the accountant for Garys, verifies receipt of merchandise and then prepares, signs, and mails the check to...
-
Increased household wealth will most likely cause an increase in: A. household saving. B. investment expenditures. C. consumption expenditures.
-
If a well-behaved investment alternative's internal rate of return (IRR) is equal to MARR, which of the following statements about the other measures of worth for this alternative must be true? 1....
-
At September 30, 2012, the accounts of Mountain Terrace Medical Center (MTMC) include the following: Accounts receivable . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . $...
-
If your BN CO wants to fund an activity scheduled to occur in November 2023, and today is February 16, 2023, who would be the best person to advise on this activity? S4, S6, or S8? Also, if the CO...
-
You have recently been hired by Keafer Manufacturing to work in its newly established treasury department. Keafer Manufacturing is a small company that produces highly customized cardboard boxes in a...
-
A company had in 2010 costs of 6,400,000, revenue of 10,000,000. In addition they repaid debt to the amount of 3,000,000. What was the profit for the 2010? Present your answer without thousand...
-
how to answer this Topic: What I Know Topic: ENDORSEMENT MINUTES K-W-L Chart What I Know What I Want to know K-W-L Chart What I Want to know What I Learned www.com What I Learned
-
2. Consider the recurrence T(n) = 2T(n/2) + f(n) in which f(n) = { n3 if [log(n)] is even n otherwise (a) Prove that T(n) = O(n) by induction. logb (b) Show that f(n) = N(n86(a)+) for some > 0 and...
-
You identify several material weaknesses over internal controls at your client, each of which resulted in a material misstatement. Management corrected all known material misstatements. Your opinion...
-
If someone asked you which bidding strategies they can use in a Standard Shopping campaign, what would you tell them? Choose two. 1.Target return on ad spend (tROAS) 2.Enhanced cost-per-click (eCPC)...
-
The maximum earned income credit prior to applying a phaseout is the (greater/lesser) of the taxpayer's earned income or the maximum earned income eligible for the credit based on their number of...
-
Herman Company has three products in its ending inventory. Specific per unit data at the end of the year for each of the products are as follows: Cost Replacement cost Selling price. Selling costs...
-
d) For die casting processes: 1. What are the most common metals processed using die casting and discuss why other metals are not commonly die casted? 2. Which die casting machines usually have a...
-
Which would you expect to be the stronger acid? Explain your reasoning in each instance. (a) CH2ClCO2H or CHCl2CO2H (b) CCl3CO2H or CHCl2CO2H (c) CH2FCO2H or CH2BrCO2H (d) CH2FCO2H or CH2FCH2CO2H
-
When, in separate reactions, 2-methylpropene, propene, and ethene are allowed to react with HI under the same conditions (i.e., identical concentration and temperature), 2-methylpropene is found to...
-
(a) Show how you might use a cyclic acetal in carrying out the following transformation: (b) Why would a direct addition of methylmagnesium bromide to A fail to give B? OH
-
You can think of a financial plan as a "financial road map to guide you through life." Develop a visual display that illustrates this concept and the five steps of the financial planning process. Try...
-
Summarize the five steps that make up the financial planning process.
-
Visit your campus career counseling office to learn about the services available to assist you with your career search and your job search. What career management services, if any, are available...

Study smarter with the SolutionInn App


