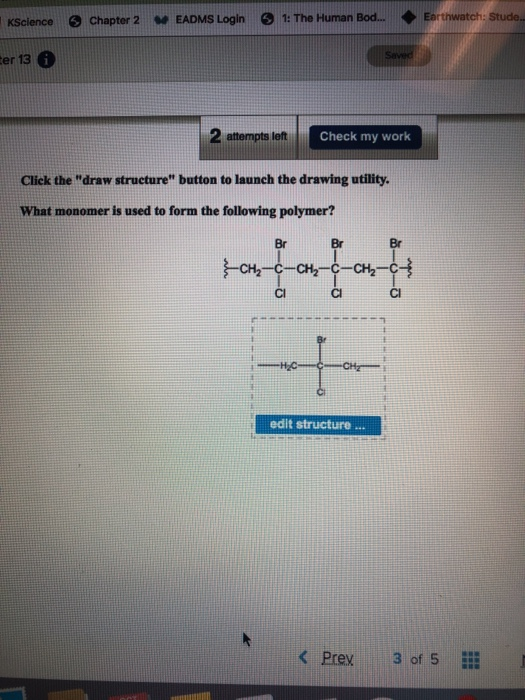
what monomer is used to form the following polym KScience ter 13 Chapter 2 EADMS Login 1:
Fantastic news! We've Found the answer you've been seeking!
Question:
what monomer is used to form the following polym
Transcribed Image Text:
KScience ter 13 Chapter 2 EADMS Login 1: The Human Bod... 2 attempts left -CH₂- Click the "draw structure" button to launch the drawing utility. What monomer is used to form the following polymer? Br Br tatat -CH₂ -CH₂ CI CI Check my work -H₂C- ta CH₂ Saved edit structure *** < Prex Br CI Earthwatch: Stude... 3 of 5 *** KScience ter 13 Chapter 2 EADMS Login 1: The Human Bod... 2 attempts left -CH₂- Click the "draw structure" button to launch the drawing utility. What monomer is used to form the following polymer? Br Br tatat -CH₂ -CH₂ CI CI Check my work -H₂C- ta CH₂ Saved edit structure *** < Prex Br CI Earthwatch: Stude... 3 of 5 ***
Expert Answer:
Answer rating: 100% (QA)
IUPAC name of monomer is 1 bromo 1 chloroethene Polymer Pol... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
What process or equipment is used to form rubber compounds into sheets?
-
What orbitals are used to form the carbon-carbon bond between the highlighted carbons? a. CH3CH==CHCH3 b. CH3CH== CHCH3 c. CH3CH == C == CH2 d. CH3== C== CCH3 e. CH3C==CCH3 f. CH2==CHCH==CH2 g....
-
The following illustration shows the orbitals used to form the bonds in carbon dioxide. Each color represents a different orbital. Label each or-bital, draw the Lewis structure for carbon dioxide,...
-
Managers should be supported through process transitions by ___________. Question 12 options: encouragement (coaching and mentoring) and assistance (necessary resource) structured approach with a...
-
Should the firm use this WACC for all projects? Explain and provide examples asappropriate. Capital structure 30% 60% 10% Cost of capital weight average Debt (bond) Common stock Preferred stock Total...
-
Fill in the blanks or answer true or false. The linear DE, y' + k 1 y = k 2 , where k 1 and k 2 are nonzero constants, always possesses a constant solution.________
-
Distinguish between a liability and an environmental liability. Is there any difference, in principle, between the recognition criteria for normal and environmental liabilities in conventional...
-
How might a developing countrys decision to reduce trade restrictions such as import tariffs affect its ability to borrow in the world capital market?
-
Using an injury scenario at work or at home that happened to you or someone you know, discuss some hard and soft barriers that, if in place, might have prevented the incident.
-
(Each part of this problem is quite long and best worked by groups of students.) Peak intensities of the molecular ion region are listed in parts (a) - (g) and shown in the figure. Identify which...
-
Linuqa (Pty) Ltd has a new product called Duel that their design team have recently developed after spending the past two years on research and development. After that long battle, the company now...
-
discuss the role of advanced quality leadership and organizational culture in fostering a quality-centric mindset, promoting employee engagement and empowerment, and creating a culture of...
-
The ledger of Queens Company at the end of the current year shows Accounts Receivable $110,000, Sales Revenue $840,000, and Sales Returns and Allowances $20,000. If Allowance for doubtful accounts...
-
First identify and label any variable(s) in this problem. (Diagram not drawn to scale.) Then write an equation(s) and show how to solve it algebraically. Be prepared to show your work. Cheryl fenced...
-
A train on a straight track sounds a 400 Hz horn when passing a parked car on the road. The train is moving at 20 m/s and the temperature is 10C. Find the apparent frequency of the horn for a person...
-
a. Construct an equal-weighted (50/50) portfolio of investments B and C. What are the expected rate of return and standard deviation of the portfolio? Explain your results.(6 Points) b. Construct an...
-
64 A city is adding a new community park on a piece of donated land. The project's baseline schedule and budget calendar is shown below. Week 3 of the project has just ended, and the project manager...
-
Prove that the mean heat capacities C P H and C P S are inherently positive, whether T > T 0 or T < T 0 . Explain why they are well defined for T = T 0 .
-
Polycarbonates are a class of thermoplastic polymers that are used in the plastic lenses of eyeglasses and in the shells of bicycle helmets. A polycarbonate is made from the re¬action of...
-
Carbon monoxide is toxic because it binds more strongly to iron in hemoglobin (Hb) than does O2. Consider the following reactions and approximate standard free energy changes:
-
The enthalpy of vaporization of chloroform (CHCl3) is 31.4 kJ/ mol at its boiling point (61.7oC). determine Ssys, Ssurr, and Suniv when 1.00 mole of chloroform is vaporized at 61.7oC and 1.00 atm.
-
List the modifications of the standard audit report that normally do not result in a qualification, a disclaimer, or an adverse opinion.
-
When more than one auditor is involved in an audit of a company's financial statements, what two decisions about reporting must the principal auditor make?
-
What disclosure is made in the principal auditors' report if they decide to assume responsibility for other auditors' work? If they decide not to assume responsibility for other auditors' work?

Study smarter with the SolutionInn App


